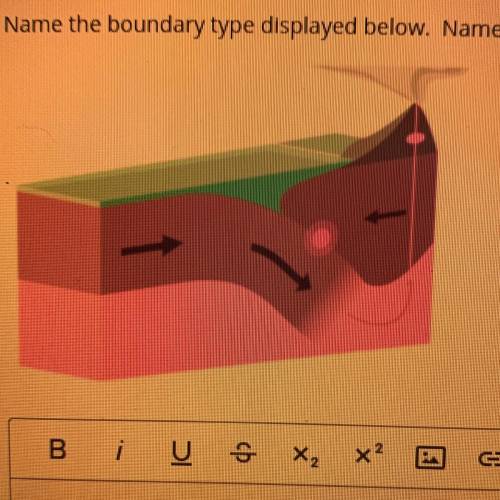
Name the boundary type displayed below. Name the types of tectonic plates that are colliding. What features are formed and why? Explain.
Solved
Show answers
More tips
- A Art and Culture The History and Characteristics of Jazz Bands: A Deep Dive...
- W Work and Career Can Skill Alone Make You a Professional?...
- F Family and Home Daughter says: If you don t want to do it, don t do it. Should we persuade her?...
- S Science and Technology How to Secure Exam Sessions: Silence Mobile Phones in the Classroom...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- P Philosophy Is Everything We Strive for Eventually Achieved and Destroyed?...
- S Society and Politics Understanding Politics and Its Role in the Development of Civilization...
- P Philosophy Why Did God Create Man and Place Him in Obscurity?...
- S Society and Politics Skoptsy: Who They Are and How They Perform Castration?...
- O Other Childhood Fears: What Many of Us Experienced...
Answers on questions: Chemistry
- C Chemistry How many milliliters of solvent is needed to make 7.840 moles of NaCl to make a 1.530 M solution ? ...
- C Chemistry To balance the reaction what coefficients (numbers) are needed: HBr +KOH --- KBr + H2O...
- C Chemistry Which of the following correctly describes an arrhenius acid...
- C Chemistry Adding salt to water increases the waters boling point...
- C Chemistry 20) marie is constructing a circuit board and observes that her current increases as she adds resistors. what statement explains why the current increases? a) she is constructing...
- C Chemistry A 7.40 L sample of N2 reacts completely at STP. the mass of the Mg consumed in the reaction is: A. 13.9 g B. 8.04 g C. 24.1 g D. 16.1 g...
- C Chemistry Convert .00000000000000123 to scientific notation alator...
- C Chemistry Multiplying scientific notation: (2.55x105). (1.23x103) =...
- C Chemistry A cube of metal weighs 1.45 kg and displaces 542 ml of water when immersed. Calculate the density of the metal....
- C Chemistry This element has two more protons than bismuth...

Ответ:
a. 2.7 mol of water
b CH2.
c. O2
Explanation:
The complete equation of the reaction should be:
2CH2(g) + 3O2(g) → 2 CO2(g) + 2 H2O(g)
a) how many moles of water will be made?
To make 2 molecules of water (H2O) we need 2 molecules of CH and 3 molecules of O2.
We have 2.7 mol of CH2, the possible yield of water produced if it all used up will be:
2.7 mol * 2/2= 2.7 mol
We have 6.3 mol of O2, the possible yield of water produced if it all used up will be:
6.3 mol * 2/3 = 4.2 mol
Since the maximum yield of CH2 lower, we can have 2.7 mol of water and have some excess oxygen at the end of the reaction.
b) What is the limiting reactant?
A limiting reactant is a reactant that will be used up in the reaction. This reactant has the lowest stoichiometric ratio compared to other reactants, which make them the one depleted out first. Since they depleted, the reaction will stop. Thus they limit the number of reactions and called limiting reactants. If you add the limiting reactant, the reaction will continue.
The limiting reactant in this reaction is the CH2. When producing water molecules, all 2.7 mol of CH2 will be used while we still have O2 left.
c) What is the excess reactant?
The excess reactant will have some remains after the reaction stop. That is because the excess reactant has more mass than needed for the reaction that will use all limiting reactants. Since we still have remains, adding excess reactant won't continue the reaction.
The excess reactant in this question is O2 since it still has remained after we make 2.7 mol of water. The O2 remaining, in this case, will be:
6.3 mol - 2.7mol * 3/2= 2.25 moles