divyaravinder8148
18.11.2020 •
Chemistry
Newton’s second law can be written using two formulas, what are they?
Solved
Show answers
More tips
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- S Style and Beauty How Are Eyelash Extensions Applied? All Your Questions Answered...
- F Family and Home Tender Care for Your Parquet: Is it Possible to Clean Parquet?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- S Sport Playing Bowling: Rules and Advice for Novices...
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
Answers on questions: Chemistry
- C Chemistry What is kinetic energy and thermal energy...
- C Chemistry During which two processes does a substance release energy...
- C Chemistry H2o2 and c6h12o6 are examples of what chemical...
- C Chemistry 3) How many L of gas is equal to 89.3 moles at STP?...
- C Chemistry 4) How many grams of F2, gas occupy 4.9 L at STP?...
- C Chemistry Calculate molecular mass of C2H5OH...
- C Chemistry If the sample has a density of 2.71 g/cm3 , how many oxygen atoms does it contain?...
- C Chemistry WILL GIVE BRAINLIEST...
- C Chemistry . Please answer number 4, I’m confused...
- M Mathematics 22% so me already learned this on class bit got suspended so i forgot...

Ответ:
In equation form, Newton's second law of motion is a=Fnetm a = F net m . This is often written in the more familiar form: Fnet = ma. The weight w of an object is defined as the force of gravity acting on an object of mass m. ... Friction is a force that opposes the motion past each other of objects that are touching.
Explanation:
have a nice day :)
Ответ:
1.24g
Explanation:
Given parameters:
Mass of titanium (IV) chloride = 4.6g
Unknown:
Mass of oxygen = ?
Solution:
To solve this problem, we need to write the balance reaction equation first;
TiCl₂ + O₂ → TiO₂ + Cl₂
Now to find the mass of the oxygen gas,
Express the given mass of the known TiCl₂ in moles:
Number of moles of TiCl₂ =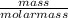
Molar mass of TiC₂ = 47.9 + 2(35.5) = 118.9g/mol
Number of moles of TiCl₂ =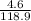 = 0.04mole
= 0.04mole
From the reaction equation;
1 mole of TiCl₂ reacted with 1 mole of oxygen gas
0.04 mole of TiCl₂ will react with 0.04 mole of oxygen gas
Mass of oxygen gas = number of moles x molar mass
molar mass of oxygen gas = 2(16) = 32g/mol
Mass of oxygen gas = 0.04 x 32 = 1.24g