stephesquilin
17.04.2020 •
Chemistry
Number 7 please I’m struggling
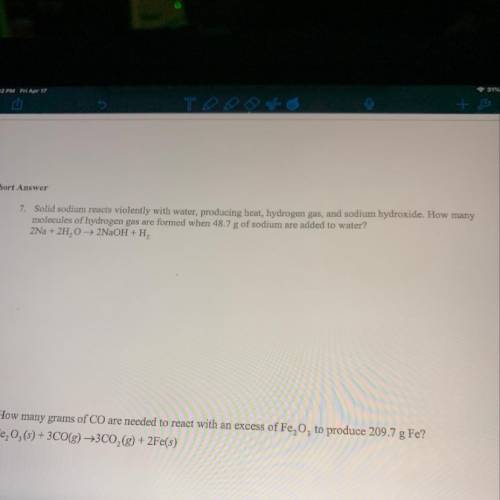
Solved
Show answers
More tips
- F Family and Home How to Build a Strong Relationship with Your Child: Tips for Effective Communication...
- H Health and Medicine Boosting Immunity: A Complete Guide on How to Improve Your Body’s Natural Defenses...
- C Computers and Internet The Best Antivirus Programs for your PC...
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
Answers on questions: Chemistry
- C Chemistry A chemist prepares a solution of barium acetate BaCH3CO22 by weighing out 52.9g of barium acetate into a 100.mL volumetric flask and filling the flask to the mark with...
- C Chemistry What is a sentence using composition....
- C Chemistry Hannnahjaxon your a f.a.g.g.o.t....
- C Chemistry F. 2.932 * 1017 H2O molecules is equal to how many grams H2O?...
- C Chemistry N2 + 3H2 → 2NH3 How many grams NH3 can be produced from 7.23 mol N2 (assuming H2 is in excess)?...
- C Chemistry 25) Which of the following would exert the most pressure on the ground? 3 points A woman standing in running shoes O A Woman standing in high heels A woman sitting...
- C Chemistry If the mass of the Crown is 505431 mg, and volume is 44 mL, what is the density of the Crown? Was the Crown solid gold? (the density of gold is 19.3 g per cubic centimetre.)...
- C Chemistry PLEASE I NEED HELP IN CHEMISTRY...
- C Chemistry I need help on this may u help me?...
- B Biology What Tropism is a bean plant growing toward a pole?...

Ответ:
Ответ:
4Al + 3O2 = 2Al2O3
To determine the mass of oxygen gas that would react with the given amount of aluminum metal, we use the initial amount and relate this amount to the ratio of the substances from the chemical reaction. We do as follows:
moles Al = 16.4 g ( 1 mol / 26.98 g ) = 0.61 mol Al
moles O2 = 0.61 mol Al ( 3 mol O2 / 4 mol Al ) = 0.46 mol O2
mass O2 = 0.46 mol O2 ( 32.0 g / mol ) = 14.59 g O2
Therefore, to completely react 16.4 grams of aluminum metal we need a minimum of 14.59 grams of oxygen gas.