On a summer day, you take a road trip through Moses Lake, WA, in a sports car. You start out at a temperature of 21°C in the morning, but the temperature in Moses Lake will reach a peak of 55°C. Each tire on your car holds 15.2 L of nitrogen gas at a starting pressure of 247 kPa. The tires will burst if the internal pressure exceeds 270 kPa.
Solved
Show answers
More tips
- P Philosophy Why Did God Create Man and Place Him in Obscurity?...
- S Science and Technology How to Make a Homemade Smoker: The Ultimate Guide...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- M Mathematics Michael earns a base pay of $500 per month plus 6% commission on his sales. What is Michael s total monthly earnings if he sells $4,000 worth of merchandise?...
- H History Which group of people was the greatest military threat to the Han dynasty?...
- M Mathematics 40.5 percent of a certain number is 65. what is the number?...
- B Biology Polar Easterlies are winds that form at the ___ and the blow east to west A.poles B.west C.east D.equator...

Ответ:
Here is the complete question.
On a summer day, you take a road trip through Moses Lake, WA, in a sports car. You start out at a temperature of 21°C in the morning, but the temperature in Moses Lake will reach a peak of 55°C. Each tire on your car holds 15.2 L of nitrogen gas at a starting pressure of 247 kPa. The tires will burst if the internal pressure exceeds 270 kPa.
a) How many moles of nitrogen gas are in each tire?
b) What would the maximum tire pressure be at 50 degrees C?
c) Will the tires burst in Moses Lake? Explain.
P = 271.9 kPa
Yes, the tires will burst in Moses Lake
Explanation:
Given that
Temperature (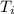 ) = 21°C = (21 +273)K
) = 21°C = (21 +273)K
= 294 K
Pressure (P) = 247 kPa
Volume (V) = 15.2 L
Numbers of moles (n)= ???(unknown)
Rate constant (R) = 0.082 L. atm/mol. K
a) How many moles of nitrogen gas are in each tire?
To determine the number of moles of nitrogen in each tire; we use the ideal gas equation:
PV = nRT
re-arranging the equation and making (n) the subject of the formula; we have:
∴ the number of moles of nitrogen (n) = 1.5384 moles
b)
b) What would the maximum tire pressure be at 50 degrees C?
at 50°C temperature
Temperature (T) = (50+273)K
= 323 K
where;
V = 1.52 L ; R = 0.082 L. atm/mol. K and n = 1.54 atm
Still using ideal gas equation:
PV= nRT
Converting the pressure from atm to KPa; we have
∴ the maximum tire pressure would be 271.9 kPa at 50 degrees C
c) Yes, the tires will have to burst in Moses Lake, because it is stated in the question that if the tire exceeds the internal pressure of 270 kPa, the tires will burst, and here we are with a pressure of 271.9 kPa. As such, the tire will definitely bursts.
Ответ:
I think your mom
Explanation:
jk jk jkjk 353.3