hayleyharding013
29.06.2019 •
Chemistry
On the basis of molecular structure and bond polarity, which of the following compounds is most likely to have the greatest solubility in water and why? a)ch4 b)ccl4 c)nh3 d)ph3
Solved
Show answers
More tips
- O Other How to Accidentally Get a Rare Coin with Your Change and How to Know Its Value?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- D Dating, Love, Relationships Why do we feel shame?...
- F Family and Home Why Does God Punish Us Constantly and How Can We Fix It?...
- F Family and Home Do Lullabies Help Babies Sleep or Is it Just a Myth?...
- B Business and Finance How to Get Your Money Back When Lending? Top 7 Ways...
- S Society and Politics Выборы: Смысл, Значение и Отражение...
- D Dating, Love, Relationships Why Should the Man be Active and the Woman Passive during Foreplay?...
- P Philosophy How did the concept of module arise in computer science?...
- S Society and Politics Why are thugs called gopniks ? A fascinating journey through Russian subculture...
Answers on questions: Chemistry
- C Chemistry Carbon-14 is used to determine the time an organism was living. The amount of carbon-14 an organism has is constant with the atmosphere, but when an organism dies the carbon-14...
- C Chemistry 420 mL of a gas at 25.0°C is compressed to 210 mL. What is the temperature of the gas after compression? Help Please ‼️‼️...
- C Chemistry Which animal can leave its eggs to hatch and does not need to take care of the infants? sea turtles ducks penguins platypus...
- C Chemistry A map titled Medieval Japan with labels A through C. The Sea of Japan is between the mainland and A. A is an island between the Sea of Japan and the Pacific Ocean. B is the mainland....
- C Chemistry 1. It serves as the geographical point of reference in determining the relation of the area the actual location of the earthquake s occurrence. a. Crust b.Focus c.Epicenter d.Wave...
- C Chemistry Consider a 50 g sample of ice at standard pressure. If it is initially at −20 ◦C and is heated until it is steam at 150 ◦C, how much total heat was added to the sample of water?...
- C Chemistry You have 100 g of pb(no3)2. how many moles is this?...
- C Chemistry What is the prime factorization of 23?...
- C Chemistry 2. cs2 what is the lewis structure? (double and triple bonds)...
- C Chemistry What s a period? what do elements in the same period have in common?...

Ответ:
The correct answer is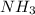 .
.
Explanation:
The electron geometry of the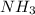 is tetrahedral and the molecular geometry is a trigonal pyramid.
is tetrahedral and the molecular geometry is a trigonal pyramid.
The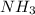 has the strongest intramolecular hydrogen bond, which makes them a highly polar molecule.
has the strongest intramolecular hydrogen bond, which makes them a highly polar molecule.
The polarity is directly proportional to the solubility of the compound in the water.
Therefore,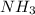 has the greatest solubility.
has the greatest solubility.
For more information, refer to the link:-
link
Ответ:
Explanation:
For each of these molecules, you must determine their VSEPR structure and then identify the strongest intermolecular forces.
Remember that water is a highly polar molecule.
a) CH₄
Electron geometry: tetrahedral
Molecular geometry: tetrahedral
Bond polarity: C-H bond nonpolar
Molecular polarity: nonpolar
Strongest IMF: London dispersion forces
Solubility in water: low
A nonpolar molecule is insoluble in a polar solvent.
b) CCl₄
Electron geometry: tetrahedral
Molecular geometry: tetrahedral
Bond polarity: C-Cl bond nonpolar
Molecular polarity: nonpolar (symmetrical molecule. All bond dipoles cancel)
Strongest IMF: London dispersion forces
Solubility in water: low
A nonpolar molecule is insoluble in a polar solvent.
d) PH₃
Electron geometry: tetrahedral
Molecular geometry: trigonal pyramidal
Bond polarity: P-H bonds are polar
Molecular polarity: polar (all P-H bond dipoles point towards P)
Strongest IMF: dipole-dipole
Solubility in water: soluble
A polar molecule is soluble in a polar solvent.
c) NH₃
Electron geometry: tetrahedral
Molecular geometry: trigonal pyramidal
Bond polarity: N-H bonds are highly polar
Molecular polarity: highly polar (all N-H bond dipoles point towards N)
Strongest IMF: hydrogen bonding
Solubility in water: highly soluble
NH₃ is so polar that it can form hydrogen bonds with water.
Ответ:
c
Explanation: