HistoryNerd7225
17.09.2021 •
Chemistry
Picture below please help it’s due in 5 minutes
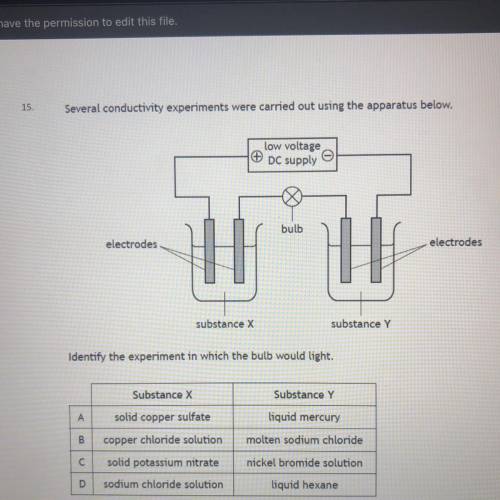
Solved
Show answers
More tips
- H Health and Medicine Where is the appendix located?...
- S Style and Beauty How to Properly Tie a Tie: 5 Simple Steps...
- L Leisure and Entertainment How to Make a Crab Trap in Just a Few Minutes...
- H Health and Medicine How Much Does Abortion Cost? Expert Answers and Insights...
- S Sport How to Build Arm Muscles? Effective Exercises and Tips...
- H Health and Medicine When can it be said that a person has a normal pulse?...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
Answers on questions: Chemistry
- C Chemistry Does ccl4 have a dipole....
- C Chemistry Two students are discussing weathering and erosion factors. Leah says that both weathering and erosion involve wind, water, and ice. Justin says that only weathering involves...
- C Chemistry If you mix 100 g of water at 10 C with 100 g of water at 30 C, what will be the equilibrium temperature...
- C Chemistry 1. A sample of a hydrocarbon is burned completely and produces 22 g of CO2 and 27.0 g of H2O. The empirical formula of the hydrocarbon is:...
- C Chemistry Anyone wannna be friends...
- C Chemistry In this experiment you need to examine the idea of thermal energy transfer using a controlled experiment what my a good question about the variables that affect derma energy...
- C Chemistry hey i really need can anyone i did attached it for you and it is since...
- C Chemistry Aneutron breaks down to form ? a. a proton and an electron b. two protons c. a helium nucleus d. an alpha particle what particle decomposes to produce the electron of beta...
- C Chemistry Which of the following is equal to 3.3 mm^3...
- C Chemistry Ablock of material has a volume of 2m^3. it has a mass of 1 ×10^4 kg. calculate the density of the material sample and express your results in g/cm^3 with appropriate scientific...

Ответ:
= 49.674 g NaCl
Explanation:
From the equation;
1 mole of Sodium metal produces 2 moles sodium chloride
This means;
23 g of Na will produce 116.88 g of NaCl
Therefore;
11.5 g will generate;
= (11.5 × 116.88)/23
= 58.44 g of NaCl
But;
Percentage yield = (Actual yield/Theoretical yield)× 100%
85 /100 = Actual yield /58.44 g
Thus;
Actual yield = 0.85 × 58.44
= 49.674 g NaCl