videogamer1192
07.12.2020 •
Chemistry
Please help me understand this.
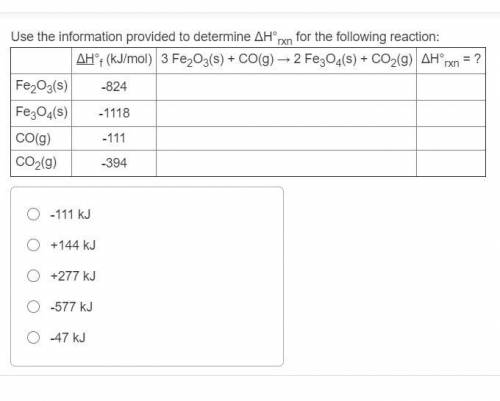
Solved
Show answers
More tips
- S Style and Beauty How to braid friendship bracelets?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- C Computers and Internet How to Learn to Type Fast?...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- D Dating, Love, Relationships How to Overcome Jealousy: Tips and Tricks...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Start Your Own Business: Tips and Recommendations...
- F Food and Cooking How to Make Delicious Cabbage Pies: The Best Recipes!...
- F Food and Cooking Discover Delicious Recipes You Can Make with Ground Meat...
- S Sport How to Do Push-ups Correctly?...
Answers on questions: Chemistry
- C Chemistry Where does the actual gas exchange happen? a. alveoli b. bronchi c. bronchioles d. cilia...
- M Mathematics Can anyone help me out?? doesn’t seem to work idk y...
- P Physics The position of a 50 g oscillating mass is given by x(t)=(2.0cm)cos(10t−π/4), where t is in s. If necessary, round your answers to three significant figures. Determine: a)...
- P Physics A solid sphere rolls up an inclined plane of inclination angle 35 degrees. At the bottom of the incline, the center of mass of the sphere has a translational speed of 13...
- E English Please read To Fly and My Brothers Flying Machine before completing these questions. They are found in the weekly checkpoint unit. How is the passage mainly organized? Question...
- M Mathematics Which shows the list below in order from greatest to least. 3.4848, -3 19/20, √11, -3.58, 362%...

Ответ:
The correct question is as follows: 0.500 moles of potassium oxide is dissolved in enough water to make 2.00 L of solution. Calculate the molarity of this solution (plz help!)
The molarity of this solution is 0.25 M.
Explanation:
Molarity is the number of moles of a substance divided by volume in liter.
As it is given that there are 0.5 moles of potassium oxide in 2.00 L of water so, the molarity of this solution is calculated as follows.
Thus, we can conclude that molarity of this solution is 0.25 M.