peytontanner
20.10.2020 •
Chemistry
Please help will give brainliest
Solved
Show answers
More tips
- G Goods and services How to Make Soap at Home: Useful Tips for Beginners...
- C Computers and Internet How to Reinstall Windows: A Detailed Guide for Beginners...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
- F Food and Cooking Лечо: вкусное и простое блюдо для любой кухни...
- H Health and Medicine Relieving Swelling in Legs: Causes and Ways to Alleviate the Symptom...
Answers on questions: Chemistry
- C Chemistry Which of the following board certified professionals must also be a medical doctor? A.psychiatric technician B.forensic social worker C.forensic psychiatrist D.forensic...
- C Chemistry If either Reaction A or B does not obey the law of conservation of mass (matter), draw additional molecules on the reactant and/or product side in order to make the...
- C Chemistry What do an atom ion and isotope have in common?...
- C Chemistry If 0.300 mol of a nonvolatile nonelectrolyte are dissolved in 3.40 mol of water, what is the vapor pressure PH2O of the resulting solution? The vapor pressure of pure...
- C Chemistry According to the ph scale, which substance is more basic than soap...
- C Chemistry Given the equation below, determine the number of moles O2 required to completely react with 28 moles of CO....
- C Chemistry Enter your answer in the provided box. What is the pH of 150.0 mL of an aqueous solution containing 0.661 g of HCI?...
- C Chemistry When copper (II) chloride reacts with sodium nitrate, NaNO3, copper (II) nitrate, Cu(NO3)2, and sodium chloride are formed. Write a balanced chemical equation for this...
- C Chemistry Which statement best describes boiling point elevation?...
- M Mathematics Unit 4 solving inequalities unit Perfolio...

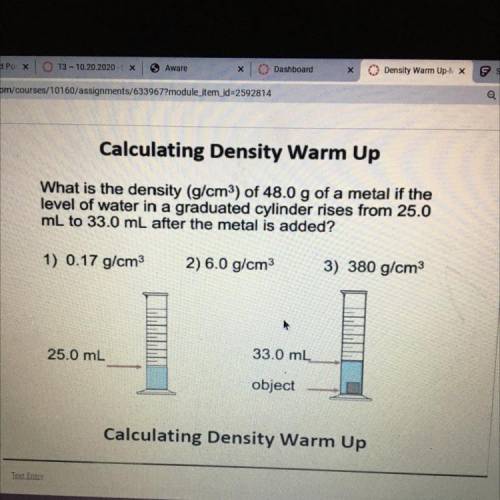
Ответ:
6 g/cm³
Explanation:Hi there !
density formula
d = m/V
V = 33.0ml - 25.0ml = 8.0 ml = 8 cm³
m = 48.0 g
replace
d = 48g/8cm³ = 6 g/cm³
Good luck !
Ответ:
Answer is: the missing pressure is 1088.66 mmHg.
Gay-Lussac's Law states that the pressure of a given amount of gas held at constant volume is directly proportional to the Kelvin temperature.
p₁/T₁ = p₂/T₂.
p₁ = 960 mmHg; pressure of the gas.
T₁ = 100°C + 273.15.
T₁ = 373.15 K; temperature of the gas.
T₂ = 150°C + 273.15.
T₂ = 423.15 K.
p₂ = p₁T₂/T₁.
p₂ = 960 mmHg · 423.15 K / 373.15 K.
p₂ = 1088.66 mmHg.