andrea1704
28.03.2021 •
Chemistry
Poor water supplies rank as a —————- in causes of death.
A. not important
B. Minor
C. Major
D. Interesting
Solved
Show answers
More tips
- G Goods and services How to Choose a Vacuum Cleaner That Will Suit You?...
- F Food and Cooking Discover the most delicious spaghetti with these tips...
- P Philosophy How did the concept of module arise in computer science?...
- F Food and Cooking How to Cook Julienne? Recipes and Tips...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- F Food and Cooking Apple Cider Vinegar: The Ultimate Health and Beauty Solution...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
Answers on questions: Chemistry
- M Mathematics The table shows different geologic time periods: period number of years ago jurassic 2.08 ⋅ 108 silurian 4.38 ⋅ 108 tertiary 6.64 ⋅ 107 triassic 2.45 ⋅ 108 order the time periods...
- C Chemistry When one fission reaction instigates exactly one more fission reaction it is called?...
- M Mathematics The table shows conversions of common units of capacity which expression can be used to find the number of liters in 15 quarts...
- E English I am doing unit test review and need help. I GIVE In a text, a fact is a piece of information that is beautifully or widly described expresses a thought or belief can be proven...

Ответ:
Because without water you will die of starvation and it is important but not many people die from it or it is C. It depends.
Hope that helps!
Ответ:
The answer to your question is Molarity = 0.0708
Explanation:
Data
NaOH 0.05 M Volume 1 = 3.87 ml Volume 2 = 25.11 ml
HCl 15 ml
Process
1.- Find the volume used of NaOH
25.11 - 3.87 = 21.24 ml = 0.02124 l
2.- Write the balanced equation of the reaction
NaOH + HCl ⇒ NaCl + H₂O
3.- Calculate the moles of NaOH in the solution
Molarity =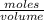
moles = Molarity x volume
moles = 0.05 x 0.02124
moles = 0.001062
4.- From the reaction we know that NaOH and HCl react in a proportion 1:1.
1 mol of NaOH ------------- 1 mol of HCl
0.001062 moles of NaOH ------------ x
x = (0.001062 x 1) / 1
x = 0.001062 moles of HCl
5.- Find the molarity of HCl
Molarity =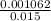
Molarity = 0.0708