ruddymorales1123
27.06.2019 •
Chemistry
Predict whether or not a precipitate will form upon mixing 175.0 ml of a 0.0055 mkcl solution with 145.0 ml of a 0.0015 m agno3 solution. identify the precipitate, if any. express your answer as a chemical formula. enter noreaction if no precipitate is formed.
Solved
Show answers
More tips
- C Computers and Internet Отправляем смс через интернет: легко и просто...
- L Leisure and Entertainment The Best Film of 2010: A Look Back at the Academy Awards...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- S Style and Beauty Autotanning: Harmful or Safe?...
- F Food and Cooking 10 Ideas for a Wedding Anniversary Gift...
- H Health and Medicine How to Reduce Sweating in the Heat and Beyond: Say Goodbye to Excessive Sweat...
- F Food and Cooking Do Aphrodisiacs Really Work? Separating Fact from Fiction...
- H Health and Medicine What to Eat to Lose Weight?...
Answers on questions: Chemistry
- C Chemistry Which is a drawback of agricultural technology? A. More food is produced B. Some dangerous chemicals are used. C. Some pests are used D. People can communicate better....
- C Chemistry What happens when you put metal into hot galium?...
- C Chemistry Find odd man out:zinc, iron, sodium, phosphorus...
- C Chemistry Is group one (alkali metals) metal, nonmetal, or metalliod?...
- C Chemistry What two forces act in the nucleus to create a nuclear tug of war ?...
- C Chemistry Which factor is responsible for the weak attraction between diatomic molecules of the halogens...
- C Chemistry What is the net ionic equation of the reaction of mgso4 with bacl2? express your answer as a chemical equation?...
- C Chemistry A farmer wants to properly manage her soil to prevent a situation like the Dust Bowl. What is the best way to manage the soil?(1 point) frequently dig the soil to get rid...
- C Chemistry What pressure, in atm, would be exerted by 0.023 grams of oxygen (O2) if it occupies 31.6 mL at 91 °C?...
- M Mathematics (1/6)^p = 36^3 x 6^18....

Ответ:
"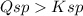 " precipitate will form. A further explanation is below.
" precipitate will form. A further explanation is below.
According to the question,
We can say the,
AgCl is a precipitate.
Ksp of AgCl,
then,
→![Qsp = [Ag^+][Cl^-]](/tpl/images/0022/4288/07ba1.png)
By substituting the values, we get
→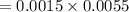
→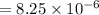
Thus the above answer is appropriate.
Learn more:
link
Ответ:
AgCl
Step-by-step explanation:
The equation for a possible reaction is
KCl(aq) + AgNO₃(aq) ⟶ KNO₃ + AgCl
The possible precipitate is silver chloride.
Moles KCl = 0.1750 × 0.0055/1
Moles KCl = 9.62 × 10⁻⁴ mol
Moles AgNO₃ = 0.1450 × 0.0015
Moles AgNO₃ = 2.18 × 10⁻⁴ mol
Total volume = 175.0 + 145.0
Total volume = 320.0 mL
[KCl] = 9.62 × 10⁻⁴/0.3200
[KCl] = 3.01 × 10⁻³ mol·L⁻¹
[AgNO₃] = 2.18 × 10⁻⁴/0.3200
[AgNO₃] = 6.80 × 10⁻⁴ mol·L⁻¹
The equation for the equilibrium is
AgCl ⇌ Ag⁺ + Cl⁻
I/mol·L⁻¹: 6.80 × 10⁻⁴ 3.01 × 10⁻³
Q_sp = [Ag⁺][Cl⁻]
Q_sp = 6.80 × 10⁻⁴ × 3.01 × 10⁻³
Q_sp = 2.04 × 10⁻⁶
K_sp = 1.8 × 10⁻¹⁰
Q_sp > K_sp, so a precipitate will form.
Ответ: