Prediction: The Cucl, crystal will dissolve in water. If we write the solvation process as a chemical
equation, we can predict two possible outcomes:
a. Cuci
H2O Cu?* (aq) + 2 Cl(aq)
H2O Cu2+ (aq) + Cl2(9)
+ 9
.
b. Cuciz
In equation (a) Cl' ions remain dissolved in water whereas in equation (b) the chlorine molecule CI,
forms and leaves the solution as a gas. Which outcome do you expect (a or b)? Explain your reasoning.
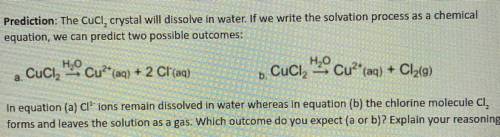
Solved
Show answers
More tips
- A Auto and Moto How to Start a Diesel Engine in Cold Weather?...
- F Family and Home How to Remove Tar Stains: Tips and Recommendations from Experts...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- S Sport How to Do a Jumping Split...
- H Health and Medicine How Did Inna Lose Weight on Dom 2?...
- F Family and Home How to Properly Fold Napkins in a Napkin Holder?...
- F Food and Cooking How to Set Up Ventrilo - The Ultimate Guide...
- S Science and Technology How to Make a Homemade Smoker: The Ultimate Guide...
- A Auto and Moto Battle for the Relocation of The Cherkizovsky Market: Who Won?...
- C Computers and Internet How Do You Refill Cartridges?...
Answers on questions: Chemistry
- E English Which of the following would BEST describe the following quote from Beatty: “The bigger your market, Montag, the less you handle controversy, remember that! All the minor minor...
- B Biology Pick three correct statements about enzymes. pls help....
- S Social Studies Some Enlightenment thinkers felt that individuals preserving current resources and transforming them into assets was a good thing for society. Part of the thinking was that...
- B Business arup works on the principle that the __economy aims to recast the way we produce and consume the world around us, keeping materials at their highest value for longer, finding...
- H History What was significant about mauryan emperor asoka...

Ответ:
Extensive properties depend on the amount of matter present, for example, the mass of gold. Intensive properties do not depend on the amount of matter present, for example, the density of gold. Heat is an example of an extensive property, and temperature is an example of an intensive property.
Explanation:
Mark brain list if you want and leave a thanks