Propanoic acid has a boiling point of 141 °c, propanamide has a boiling point of 213 °c, and propanal has a boiling point of 48 °c. rationalize the differences in boiling point between these three compounds.
Solved
Show answers
More tips
- O Other What happens if you get scared half to death twice?...
- F Family and Home What s That Noise When a Kettle Boils? The Science of Water and Steam...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- W Work and Career How much does an honest traffic police officer earn in a day?...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
- H Health and Medicine Is Massage Necessary? Facts and Opinions...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet Best Antivirus: How to Choose Protection for Your Computer?...
- F Food and Cooking When is Easter in 2011?...
Answers on questions: Chemistry
- C Chemistry 5 point How many molecules are in 50g of CO2?...
- C Chemistry Which compound reacts with an acid to form a salt and water?...
- C Chemistry Which element in period 3 has both metallic and non metalik properties...
- C Chemistry Which of these is not an example of chemical weathering? a. iron-rich mineral rusting b. feldspar turning into clay c. limestone reacting with acid d. granite breaking up into...
- M Mathematics I’ll give brainliest....
- H History Was gold a major export of the English colonies...
- S Social Studies How do sociologists seek to explain the experience of urban life?...
- H History What was one of the major political developments in Europe during the Late Middle Ages? A. the establishment of feudal manors in rural areas B. the establishment of civic government...
- E English How is dialogue in a story different from dialogue in real life?...
- M Mathematics Order the following from least to greatest. * 34 - 16 0 - 41 12...

Ответ:
Explanation:
As it is given that boiling point of propanamide is very high. So, reason for this is that easy formation of hydrogen bonds which are strong enough that we have to provide large amount of heat to break it.As in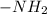 , the hydrogen atoms which are present are positive in nature. Due to this they are able to form hydrogen bonds with the neighboring oxygen atom.
, the hydrogen atoms which are present are positive in nature. Due to this they are able to form hydrogen bonds with the neighboring oxygen atom.
Hence, these bonds are so strong that high heat needs to given to break them.
A propanoic acid contain carboxylic group as the functional group. So, this group is also able to form hydrogen bonding as it forms a hydrogen bond between an acid group and hydroxyl group of neighboring molecule.Hence, it will also require high heat to break the bond due to which there will be increase in boiling point.
In propanal, there is presence of aldehyde functional group and three carbon atoms chain which will not form strong bonding with the hydrogen atom of CHO. Due to this there will exist weak Vander waal's force that is not at all strong enough.As a result, less energy will be needed to break the bonds in propanal. Hence, it has very low boiling point.
Ответ:
the stoichiometry in this case of H₂ to O₂ is 2:1.
This means that 2 moles of H₂ reacts with 1 mol of O₂.
we have been asked to calculate the volume of H₂ gas.
Since both O₂ and H₂ are gases, at standard conditions its stated that molar volume of gases is 22.4 L
This means that at standard temperature and pressure, 1 mol of any gas occupies a volume of 22.4 L
first we need to calculate the number of O₂ moles reacted;
22.4 L of gas - 1 mol of O₂
1 L of gas - 1/22.4 mol/L
then 20 L of O₂ - 1/22.4 mol/L * 20 L = 0.89 mol
stoichiometry of H₂ to O₂ is 2:1
the number of H₂ moles = 0.89*2 = 1.79 mol
1 mol occupies 22.4 L
Therefore 1.79 mol = 22.4 L/mol * 1.79 mol = 40 L
Therefore it can be seen that stoichiometry applies to volumes as well.
volume of H₂ : O₂ = 40 L : 20 L = 2:1
volume and moles both can be determined by stoichiometry.
Volume of H₂ reacted = 40 L