ismailear18
07.03.2021 •
Chemistry
Propose a mechanism for the following reaction
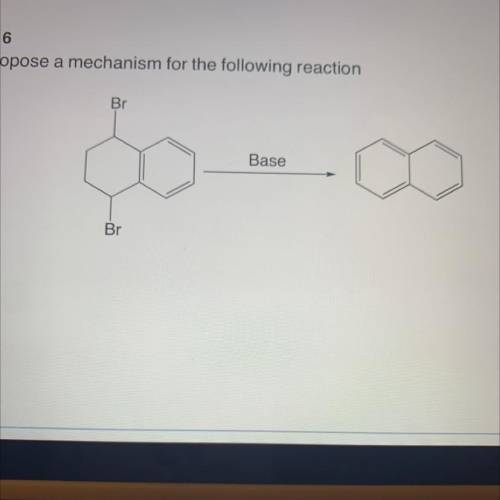
Solved
Show answers
More tips
- G Goods and services Stock center - a modern way of optimizing logistics...
- F Food and Cooking How to Properly Wash a Down Jacket? Tips from Experts...
- C Computers and Internet Thin Client: What It Is and Why You Need It?...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- C Chemistry How many molecules are contained in 201 g of magnesium sulfate heptahydrate?...
- C Chemistry I need help ASAP pls...
- C Chemistry Please help me understand how to... Calculate the mass of Cr2O3 that can be produced if the reaction of 56.2 g of chromium and sufficient oxygen has a 76.0 % yield. ...Thank...
- C Chemistry Determine the temperature of the gas with a pressure of 2.5 ATM and a volume of 15 L. Assume R is 0.0821 ATM*L/mol*K A)91.35 KB)91.35 CC)7.5 KD)7.5 C...
- C Chemistry If liquid A has a density of 0.600g/mL, liquid B has a density of 1.00 g/ml, and liquid C has a density of 0.520g/mL, which liquid would be on the top layer if they were...
- C Chemistry Santa maria has an elevation of 6.30 times 10 to the 5th mm. how many km is the elevation?...
- C Chemistry Areaction was performed in which 4.0 g of cyclohexanol was reacted with an acid catalyst to obtain 2.8 g of cyclohexene. calculate the percent yield for this reaction....
- C Chemistry Movement of solute from a 0.2% solute concentration solution to a 0.4% solute concentration solution is an example of...
- C Chemistry Which of the following is not the same as 105.6 milligrams? 0.1056 g 0.0001056 kg 0.001056 dkg...
- C Chemistry Complete the lab below using household items and submit your responses to the analyze and conclude questions as a portfolio item on the assessment page of this lesson....

Ответ:
i dont know the answer
Explanation:
when i get the answer i will say you
Ответ:
See explanation
Explanation:
You see, we must cast our minds back to Charles' law. Charles' law gives the relationship between the volume of a gas and temperature of the gas.
Now, Micheal left the balloon outside at a particular temperature and volume the previous night. Overnight, the temperature dropped significantly and so must the volume of the gas in the balloon!
Remember that Charles' law states that, the volume of a given mass of gas is directly proportional to its absolute temperature at constant pressure. Since the pressure was held constant, the drop in the volume of gas in the balloon can be accounted for by the drop in temperature overnight.