goreeefk5205
17.08.2020 •
Chemistry
Question 14 (5 points)
What's the acid ionization constant for an acid with a pH of 2.11 and an equilibrium
concentration of 0.30 M?
O A) 4.87x10-8
B) 1.99x10-6
C) 3.32x10-4
OD) 2.01x10-4
Solved
Show answers
More tips
- W Work and Career 10 Best Ways To Find A Job: Tips To Land Your Dream Job...
- L Leisure and Entertainment How to Choose a Program for Cutting Music?...
- A Auto and Moto How to choose the right drive for your BMW...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
Answers on questions: Chemistry
- C Chemistry What is the rate law of NO2 gas...
- C Chemistry What sort of things can we identify using a ditchotomous key?...
- C Chemistry How many moles of carbon dioxide are produced when 5.12 moles of glucose undergoes a combustion reaction?...
- C Chemistry The formation of which compound (from its elements) releases the greatest amount of energy per mole at 298 Kand 101.3 kPa? * CO2 (g) C2H4 (g) C2H6 (g) C2H2 (g)...
- C Chemistry Identify the number of core and valence electrons for each atom. 1s22s22p3 core electrons and valence electrons 1s22s22p63s23p5. core electrons and. valence electrons...
- C Chemistry calcium hydroxide is commonly known as agricultural lime and is used to adjust the soil ph. before the lime was added to a field, the soul ph was 5. after the lime was added,...
- C Chemistry Consider the following equation. h2o(l)+heat - h2o(g) which will occur of the if the temperature of the system is increased...
- C Chemistry Explain How do muscles work in pairs to move the body?...
- M Mathematics While shopping at a music store, Rick bought 7 CD s, all at the same price. The tax was $6.50 and the total was $66.00. What was the price of each CD?*...
- C Chemistry • Anything that has mass and takes up space PLS HELPPPP...

Ответ:
D) 2.01 x 10⁻⁴ .
Explanation:
pH = 2.11
[ H⁺ ] =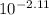
Let the acid be HA
It will ionise as follows .
HA ⇄ H⁺ + A⁻
in equilibrium .30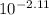
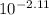
Acid ionisation constant Ka =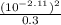
= 2 x 10⁻⁴
Ответ:
increase the temperature from 27 degrees celsius to 37 degrees celsius
Explanation;The rate of an enzyme-catalyzed reaction increases as the temperature is raised.The human body is maintained at 37°C as this is the temperature at which the enzymes in our body work best. Therefore increasing the temperature from 27 degrees Celsius to 37 degrees Celsius increases enzyme activity and in turn increases the production of amino acid in the flask.Higher temperatures than 37° C which is the optimum temperature, disrupt the shape of the active site, which will reduce enyzme activity, or prevent it from working, in other words the enzyme will have been denatured.