bettybales1986
12.03.2022 •
Chemistry
Raj needs to determine the chemical composition of trace evidence like paint and fiber. Which tool would Raj use to do this?
comparison microscope
scanning electron microscope
microspectrophotometer
gas chromatograph
Solved
Show answers
More tips
- C Computers and Internet Best Applications for Your iPad: Review of the Best Candidates for Installation...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
Answers on questions: Chemistry
- C Chemistry Convert 3.99/gallon to dollars per liter...
- C Chemistry A solution is made by dissolving 37.5 g of sodium sulfide (Na2S) in 217 g of water. What is the molality of the solution?...
- C Chemistry Question 6 of 10 How does the scientist benefit society?...
- C Chemistry A copper pipe contains 21,000 g of copper. How many moles of copper are in the pipe?...
- C Chemistry PLS HELPPPP!! I need hellppp...
- C Chemistry 3. An element has two isotopic forms. One has a mass of 37 amu and one has a mass of 40 amu. If the average atomic mass of the element is 39 amu, what can be stated about the relative...
- C Chemistry Can somebody help me with my career work please?...
- C Chemistry How many atoms in the following below. FeS2 - Na2S2O3...
- C Chemistry CO2+H2O- H2CO3 Balancear la ecuación...
- C Chemistry What volume of a 0.385 M hydrochloric acid solution is required to neutralize 10.4 mL of a 0.159 M calcium hydroxide solution...

Ответ:
4NH3 + 3O2 → 2N2 + 6H2O
a. 2 moles
b. 4 moles
c. 3 moles
Explanation:
a. 4 mol NH3
4NH3 + 3O2 → 2N2 + 6H2O
4 : 3 : 2 : 6
4 (mol)
⇒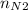 = 4 × 2 ÷ 4 = 2
= 4 × 2 ÷ 4 = 2
b. 4 mol N2
c. 4.5 mol O2
4NH3 + 3O2 → 2N2 + 6H2O
4 : 3 : 2 : 6
4.5 (mol)
⇒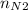 = 4.5 × 2 ÷ 3 = 3
= 4.5 × 2 ÷ 3 = 3