AnaiyaKirksey8
28.09.2019 •
Chemistry
Read the statement.
a 200 ml nacl solution with a concentration of 4.0 g/l is mixed with a 600 ml solution containing 8% nacl (m/v).
what is the final concentration of salt in the solution in g/l?
a.4.7
b.61
c.6.1
d.47
Solved
Show answers
More tips
- G Goods and services How to Choose a Coffee Maker? The Ultimate Guide for Coffee Lovers...
- O Other How to Accidentally Get a Rare Coin with Your Change and How to Know Its Value?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- D Dating, Love, Relationships Why do we feel shame?...
- F Family and Home Why Does God Punish Us Constantly and How Can We Fix It?...
- F Family and Home Do Lullabies Help Babies Sleep or Is it Just a Myth?...
- B Business and Finance How to Get Your Money Back When Lending? Top 7 Ways...
- S Society and Politics Выборы: Смысл, Значение и Отражение...
- D Dating, Love, Relationships Why Should the Man be Active and the Woman Passive during Foreplay?...
- P Philosophy How did the concept of module arise in computer science?...
Answers on questions: Chemistry
- C Chemistry Brainliest pl Erosion and weathering are synonyms True or false...
- C Chemistry Need help w chemistry pls...
- C Chemistry 4. Which renewable resource generates the greatest amount of energy? sc.7.P.11.2 A biomass ® coal © hydroelectric D natural gas...
- C Chemistry Hydrogen combines with oxygen to form water. How many moles of water are produced when 5.00 moles of oxygen are used?...
- C Chemistry ASAP SOS What are the spectator ions for the following reaction? Ba(ClO3)2 + (NH4)2SO4 — BaSO4 + 2NH4ClO3a. Ba^2+, SO4^2-b. Ba^2+, NH^4+c. ClO^3-, SO4^2-d. NH^4+, ClO^3-...
- C Chemistry Suppose the irregularly shaped object is a massive whale shark that is about to be moved into a cube-shaped aquarium tank. how might you combine the “length x width x height” formula...
- C Chemistry Calculate the actual mass of water that will form if 20.9 of n2H4 reacts with 51.4g of oxygen gas...
- M Mathematics What is the rate of change of y with respect to x for this function?...
- M Mathematics Use matrices to determine the coordinates of the vertices of the rotated figure. Then graph the pre-image and the image of the same coordinate grid....
- C Chemistry 3. When a solution of sodium hydroxide is mixed with iron (II) chloride, a green precipitate is formed. What is the balanced equation for that reaction? O Fe2Cl(aq) + NaOH(aq) → Fe2OH(aq)...

Ответ:
Answer : The correct option is, (B) 61 g/L
Explanation :
First we have to calculate the mass of NaCl.
As, 1000 ml NaCl solution contain 4 grams of NaCl
So, 200 ml NaCl solution contain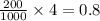 grams of mass of NaCl
grams of mass of NaCl
Now we have to calculate the mass of another NaCl.
As, 100 ml NaCl solution contain 8 grams of NaCl
So, 600 ml NaCl solution contain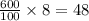 grams of NaCl
grams of NaCl
Total mass of NaCl = 0.8 + 48 = 48.8 g
Total volume of NaCl solution = 200 + 600 = 800 ml
Now we have to calculate the final concentration of salt in the solution.
Therefore, the final concentration of salt in the solution is, 61 g/L
Ответ: