lllmmmaaaooo
10.03.2020 •
Chemistry
Select the correct answer.
In which type of chemical reaction is heat absorbed from the surroundings?
A. endothermic
B. irreversible
C. reversible
D. decomposition
OE disintegration
Reset
Next
Solved
Show answers
More tips
- C Computers and Internet Boost your processor performance with these easy tips...
- L Leisure and Entertainment When will Maslenitsa start?...
- F Food and Cooking Discovering the Mysterious Fruit of Feijoa...
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
Answers on questions: Chemistry
- S Social Studies All of the following were true as a result of rural to urban migration in Texas during the 20th century EXCEPT: F fewer family farms G older rural population H larger corporate...
- E English Write a short paragraph that explains the central idea of the article Bolt is world s fastest runner. Use at least two details from the article to support your response....

Ответ:
Explanation:
The given data is as follows.
Density of vinegar = 1.0 g/ml
Specific heat capacity = 4.25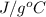
Relation between enthalpy and specific heat is as follows.
Hence, putting the values into the above formula as follows.
= (as density =
(as density = 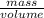 )
)
= - 315 J
Thus, we can conclude that the enthalpy of reaction is -315 J.
As the value is negative so, it means that heat is releasing. Hence, the reaction is exothermic in nature.