mikaelalcool1
28.10.2020 •
Chemistry
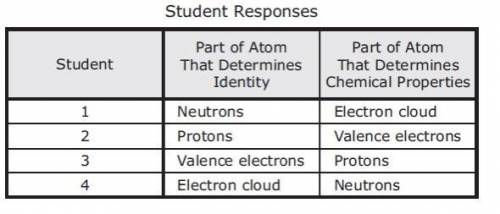
Several students are describing the different parts of the atoms. Which student is correct?
1.
2.
3.
4.
Solved
Show answers
More tips
- S Style and Beauty What to Do When Hair Starts Falling Out?...
- H Health and Medicine Novomin: What is it and how to use it?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
Answers on questions: Chemistry
- E English HAHAHA TRUMP IS GOING TO BE BACK IN OFFICE YOU WILL ALL SEE You see over 200 members of congress have been arrested including Nancy Pelosi and other high figures, you c i know this...
- M Mathematics There are 169 chairs in the gymnasium that need to be arranged in rows and columns. how can they be divided so that there are an equal amount of rows and columns?...
- B Biology Agroup of scientists are studying a crab. after comparing the crab to other crabs and testing it’s dna, the scientists think they have found a new species of crab. what should they...
- E English very , replied the mouse. how you? look at the blank with the number 5 in the passage. which of these answers correctly completes the sentence? a) am b) are c) be d) is...
- H History What was the first document to outline structure of the united states government?...

Ответ:
The chemical equation is
Explanation:
There are 2 types of reactions that are classified based on enthalpy change:
Endothermic reactionExothermic reactionEndothermic reactions: They are defined as the reactions where heat is absorbed by the reaction. The change in enthalpy of the reaction is always positive.
Exothermic reactions: They are defined as the reactions where heat is released by the reaction. The change in enthalpy of the reaction is always negative.
Given values:
Energy released for 1 mole of HCl reacted = -42.1 kCal
The chemical equation for the formation of ammonium chloride follows: