caitybugking
08.07.2021 •
Chemistry
Si 40 g de un compuesto C6H10O5 se disuelven en 500 g de agua, determine el punto de ebullición de esta solución. (Agua: temperatura de ebullición 100 °C y Ke = 0,52 °C/m)
Solved
Show answers
More tips
- W Work and Career 10 Best Ways To Find A Job: Tips To Land Your Dream Job...
- L Leisure and Entertainment How to Choose a Program for Cutting Music?...
- A Auto and Moto How to choose the right drive for your BMW...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
Answers on questions: Chemistry
- C Chemistry Will it give me the right answers...
- C Chemistry Mothballs are composed primarily of the hydrocarbon naphthalene (C10H8)(C10H8). When 1.025 gg of naphthalene is burned in a bomb calorimeter, the temperature rises from 24.25 ∘C∘C...
- C Chemistry Drag each tile to the correct location. Which type of change does the statement describe?Particles rearrange into newcombinations, forming newsubstances.There is no change in the chemicalproperties...
- C Chemistry Which equation is not balanced? 2H2 + O2® 2H20 4H2 + 202® 4H20 2H2 + O2® H20 H2 + H2 + O2® H2O + H20...
- C Chemistry A chemist dissolves 73 grams of sodium chloride (NaCl) in water. The final volume of the solution is 1,050 milliliters. What is the molarity of the solution?...
- C Chemistry PLEASE HELP ILL GIVE U BRAINLIEST What s something you learned about the periodic table...
- C Chemistry PLEASE HELPWhat do you know about valence electrons?...
- C Chemistry P4 + NaOH + H2O—— PH3 + Na2HPO3 Balance given equation by oxidation no. Method...
- C Chemistry If five elements have atomic numbers 2,3,6,9 which two numbers are the most reactive??...
- C Chemistry The molarity of concentrated acetic acid is 17.45 M. When acetic acid is diluted in water it is commonly called vinegar with a molarity of 0.83M. Lays potato chips needs to make 250....

Ответ:
The question is: If 40 g of a compound C6H10O5 are dissolved in 500 g of water, determine the boiling point of this solution. (Water: boiling temperature 100 ° C and Kb = 0.52 ° C / m).
The boiling point of given solution is .
.
Explanation:
Given: Mass of solute = 40 g
Mass of solvent = 500 g (1 g = 0.001 kg) = 0.5 kg
Molality is the moles of solute present in kg of solvent.
Moles is the mass of substance divided by its molar mass. Hence, moles of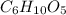 (molar mass = 162.141 g/mol) is calculated as follows.
(molar mass = 162.141 g/mol) is calculated as follows.
Now, molality of the solution is calculated as follows.
The boiling point is calculated as follows.
where,
m = molality
Substitute the values into above formula as follows.
As the boiling point of water is 100 degree Celsius. So, the boiling point of solution is as follows.
Thus, we can conclude that the boiling point of given solution is .
.
Ответ:
The explanation that the researcher can give for the fact that the color of the leaves of both plants has not changed is that receive the same radiation from the sun .
Explanation:
English:
A researcher observes two plants of the same species, but with different adaptations. Plants A have green leaves, very deep roots with very dense hairs. Plants B, on the other hand, have very shallow roots, the density of their villi is less dense, although the colour of the leaves is the same. What explanation can the researcher give for the fact that the colour of the leaves of both plants has not changed?
Option for this question:
Receive the same amount of water .Receive the same radiation from the sun .Receive the same amount of oxygen .Receive the same amount of carbon dioxide.The coloration of leaves in plants depends on the interaction of sunlight with photosynthetic pigments, such as chlorophyll, found in their leaves.
From the idea that both plants are of the same species, it can be inferred that both have the same composition of photosynthetic pigments in their leaves, the amount of sunlight they receive being the factor that makes them have the same coloration, without changes.
The adaptations they possess and which differentiate them are the roots, which are responsible for the absorption of water and nutrients from the soil, which does not influence the color of the leaves.
Learn more:
Photosynthesis in a leaf