elliswilliams1063
28.08.2019 •
Chemistry
Sodium chromate is used to protect iron from corrosion and rusting. determine its formula mass. 261.97 amu 238.98 amu 74.99 amu 161.97 amu 138.98 amu
Solved
Show answers
More tips
- C Computers and Internet What is the Meaning of lol and How Did it Become Popular?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- H Health and Medicine How to Help Men Gain Weight?...
- F Family and Home How to Build a Strong Relationship with Your Child: Tips for Effective Communication...
- L Legal consultation Juvenile Justice: Who Needs It?...
- F Food and Cooking What age is appropriate for giving your child cocoa?...
- A Animals and plants Why do cats go crazy over catnip?...
- H Health and Medicine How to Cure Adenoids?...
- S Science and Technology Why is there no gravity on other planets?...
- F Food and Cooking How to Properly Wash a Down Jacket? Tips from Experts...
Answers on questions: Chemistry
- C Chemistry Students want to gather evidence for the claim that the number of atoms present before a chemical reaction is equal to the number of atoms present after the chemical reaction....
- C Chemistry Given that you have 14.5 moles of n2, how many moles of h2 are theoretically needed to produce 22.5 moles nh3 according to the reaction n2 + 3 h2 → 2 nh3 ? 1. 21.8 moles...
- M Mathematics If a function is continuous at an x-value, does the limit always exist at the x-value. Explain why or why not....
- E English Reading Quiz Question 2 Multiple Choice Instructions Read the question carefully and select the best answer. Which of the following best explains why the ticket seller believes...
- M Mathematics Find the average rate of change of f(x)=5x2 - 3 from 3 to 9. The average rate of change of f(x) = 5x2 - 3 from 3 to 9 is (Simplify your answer.) help...
- M Mathematics Sketch the graphs -2x +3y=-1...
- C Chemistry Mark and John are lifting weights at the gym. It takes mark 45 N of force to lift one of the weights. If John only got the weight 50% of the way up, how many newtons of...
- B Biology Each enzyme has a particular substrate because enzymes...
- S SAT ashima found 2 boxes of sugar in the kitchen. the weight of green box is 1.26 kg and of the red box is 1.026 kg. which box contains more sugar and by how much?...
- C Chemistry Calculate the final molarity of H2O2 if 5.5 mL of a 3.0% w/w H2O2 solution, which has a density of 1.0 g/mL, is added to 5.5 mL of a starch-iodide solution....

Ответ:
1) Nomenclature: Chromate
Chromate is an ion (anion) from the chromic acid.
Chromic acid is formed from Chromic oxyde with the atomic number 6.
Then Cr + O2 -> Cr2O6 = CrO3
CrO3 + H2O -> H2CrO4
H2CrO4 = 2H(+) + CrO4 (2-) < this is the ion chromate
Now you have to join the chromate ion with the sodium ion => Na2 (CrO4)
2) Now you can calculate the molar mass of the formula Na2(CrO4).
You must only muliply every subscript times the atomic mass of the element:
Na2 => 2 * 23 g/mol = 46 g/mol
Cr => 52 g/mol
O4 => 4 * 16g/mol = 64 g/mol
Formula mass = 46 g/mol + 52 g/mol + 64 g/mol = 162 g/mol
I used the numbers with less significant figures thant the problem required but the answer is evident. Also, g / mol = a.m.u = atomic mass unit.
161.97 a.m.u
Ответ:
Answer : The net ionic equation will be,
Explanation :
In the net ionic equations, we are not include the spectator ions in the equations.
Spectator ions : The ions present on reactant and product side which do not participate in a reactions. The same ions present on both the sides.
The given balanced ionic equation will be,
The ionic equation in separated aqueous solution will be,
In this equation,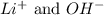 are the spectator ions.
are the spectator ions.
By removing the spectator ions from the balanced ionic equation, we get the net ionic equation.
The net ionic equation will be,