Solve this stoichiometry problem 2 2 unread replies. 2 2 replies. solve the following problem showing all of your work and including all units. you can solve the problem in several steps or as a tandem conversion problem. how many grams of br2 are needed to completely convert 15.0 g al to albr3 ? al (s) + br2 (l) --> albr3 (hint: the equation is not balanced)
Solved
Show answers
More tips
- S Science and Technology Discovering the Anatomy of an LCD TV Screen...
- F Family and Home How to Raise a Genius? Discover the Secrets Here...
- F Food and Cooking How to Properly Cook Buckwheat?...
- H Health and Medicine How to Tan in a Tanning Bed? Tips and Recommendations...
- S Style and Beauty How to Sew a Balloon Skirt: Detailed Tutorial and Tips on Choosing the Right Fabric...
- P Philosophy Personal attitude towards Confession: how to prepare and undergo the procedure correctly?...
- F Food and Cooking How to Make Cottage Cheese at Home: Simple and Quick Recipe with Step By Step Instructions...
- H Health and Medicine Which Water Are You Drinking? Is it Worth Buying Bottled Water?...
- H Health and Medicine What Makes a Man a Man?...
- H Health and Medicine What to Take with You to the Maternity Hospital?...
Answers on questions: Chemistry
- C Chemistry Which of the statements below is correct regarding the gravity of the Earth? The Earth s gravity... Group of answer choices A.Pulls on the gases in the Earth s atmosphere....
- C Chemistry Antoine Lavoisier is known for which of the following? (Select all that apply) a. the theory of general relativity b. the theory of natural selection c. the law of conservation...
- C Chemistry What is the mass percent concentration of a solution prepared from 500...
- C Chemistry Which of the following is released during cellular respiration? a.carbon dioxide b.glucose c.mitochondria d.oxygen...
- C Chemistry 3. As agitation increases, the rate at which a solute will dissolve . increases decreases...
- C Chemistry Suppose a gas diffuses 1.4 times as fast as sulfur dioxide at the same temperature and pressure. What is the molarmass of the unknown gas?...
- C Chemistry Sing pure water, set the room temperature to -10.0 °c and observe. what do you notice about the water temperature as the water is freezing? the water temperature stays constant...
- C Chemistry Express (3 x 104) (4 x 1023) in scientific notation...
- C Chemistry 4.77g of an oxide of copper contains 3.81g of copper.calculate the empirical formula for the compound...
- C Chemistry Oxit bazo kết hợp được mấy loại...

Ответ:
Answer : For, this question,
The balanced equation is; 2Al + 3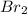 ----> 2Al
----> 2Al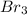
We can now, calculate how many grams of will be needed to completely convert 15 g of Al into Al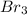 .
.
Here, the reaction produces, 2 moles of Al![Br_{3}[/tex} by consuming 2 moles of Al and 3 moles of [tex]Br_{2}](/tpl/images/0092/6201/f0357.png)
So, For 15.0 g of Al; we can calculate;
(15.0 g Al) X (1 mole Al / 26.98 g Al) X (3 moles / 2 moles Al) X (159.81 g / 1 mole )
= 133 g of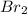 will be needed.
will be needed.
Therefore, 133g of will be needed to completely convert 15 g of Al into Al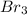 .
.
Ответ:
Answer : The mass of needed are, 131.8 grams
needed are, 131.8 grams
Solution : Given,
Mass of Al = 15 g
Molar mass of Al = 27 g/mole
Molar mass of = 159.8 g/mole
= 159.8 g/mole
First we have to calculate the moles of Al.
Now we have to calculate the moles of
The given balanced reaction will be,
From the balanced reaction, we conclude that
As, 2 moles of Al react with 3 moles of
So, 0.55 mole of Al react with of
of 
Now we have to calculate the mass of
Therefore, the mass of needed are, 131.8 grams
needed are, 131.8 grams
Ответ:
Explanation:
A prussian blue colouration indicates the presence of nitrogen in the compound.
If nitrogen is present in the compound, the sodium extract would contain sodium cyanide formed during fusion.
On adding the required reagents, sodium cyanide reacts to form ferric-ferrocyanide which has prussian blue colour.
I NEED BRAINLIEST ✌️ PLZ Itz snow