Someone help fast i will love u forever
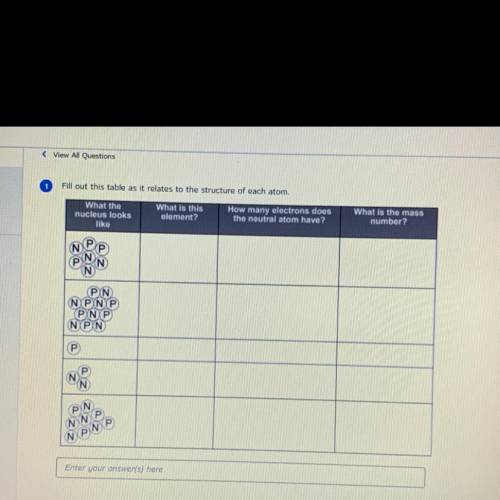
Solved
Show answers
More tips
- A Animals and plants How ants survive winter: exploring the secrets of their winter life...
- F Food and Cooking Discover How to Properly Prepare Dough for Rasstegai...
- P Philosophy Unidentified Flying Object - What is the Nature of this Phenomenon?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
Answers on questions: Chemistry
- M Mathematics What is x+y Explain your answer...
- M Mathematics Determine the simple interest applied to a principle over 20 years if the total interest paid equals the borrowed principal...
- S Social Studies In order to prove that learning has happened, it is necessary to A. measure performance objectively and concretely B. repeat each learning event at least three times C. ask the...
- M Mathematics PLS ANSWER!!! Lori graphed f(x) = −34x − 1 and g(x) = −4f(x) on the same coordinate plane. Which statements below incorrectly describe how the graphs of f(x)and g(x)are related?...
- B Business Tax incidence is the division of the burden of tax between . A. buyers, sellers, and the government B. sellers and the government C. workers and employers D. buyers and sellers...

Ответ:
Bronsted-Lowry acid
Explanation:
Bronsted-Lowry Base -
According to Bronsted-Lowry theory of acids and bases:
Acids are those substances which donate proton in the solution whereas bases are those substances which accept proton in the solution.
Consider an example,
HCO3- behave as base as it accepts proton in the reaction whereas HOH behaves as acid as it releases proton.
According to Lewis theory of acids and bases:
Acids are electron acceptor and bases are electron donar. Acids are generally electron deficient species whereas Lewis are chemical species having lone pair of electrons.
For example, AlCl3 is behave as Lewis acid because of being electron deficient species.
NH3 is a Lewis base because of presence of lone pair of electrons.
Therefore, among given options, Bronsted-Lowry acid is the correct option.