SOMEONE, PLEASE HELP ME AND ILL HELP YOU THIS TAKES MY WHOLE GRADE
What is a pure substance?
A. a material that cannot be physically separated into its parts
B. a substance that cannot be separated or broken down into simpler substances by chemical means
C. the smallest unit of an element that has the chemical identity of that element
D. an arrangement of all the known elements in order of their atomic number
#2 - TRUE OR FALSE: Smelting is a process that uses heat to separate pure metals from a mixture that also contains other substances.
A. True
B. False
#3 - TRUE OR FALSE: Currently, 200 elements are known.
A. True
B. False
#4 - What is an element?
A. a material that cannot be physically separated into its parts
B. the smallest unit of an element that has the chemical identity of that element
C. a substance that cannot be separated or broken down into simpler substances by chemical means
D. an arrangement of all the known elements in order of their atomic number
#5 - TRUE OR FALSE: Only two elements can have exactly the same set of properties
A. True
B. False
#6 - What is an atom?
A. a substance that cannot be separated or broken down into simpler substances by chemical means
B. an arrangement of all the known elements in order of their atomic number
C. a material that cannot be physically separated into its parts
D. the smallest unit of an element that has the chemical identity of that element
#7 - TRUE OR FALSE: Atoms are so tiny that they cannot be seen with an optical microscope.
A. True
B. False
#8 - TRUE OR FALSE: Gold and silver are both soft metals that react vigorously with water and air.
A. True
B. False
#9 - What is the periodic table?
A. a substance that cannot be separated or broken down into simpler substances by chemical means
B. a material that cannot be physically separated into its parts
C. the smallest unit of an element that has the chemical identity of that element
D. an arrangement of all the known elements in order of their atomic number
#10 - TRUE OR FALSE: The first column of the table contains a group of very reactive metal elements.
A. True
B.False
Solved
Show answers
More tips
- L Leisure and Entertainment Choosing the Right Books to Read: Tips and Recommendations...
- L Leisure and Entertainment What can be considered an antique: defining and valuing old objects...
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- C Computers and Internet Log-in: what is it and why do you need it?...
- C Computers and Internet What is the Meaning of lol and How Did it Become Popular?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- H Health and Medicine How to Help Men Gain Weight?...
- F Family and Home How to Build a Strong Relationship with Your Child: Tips for Effective Communication...
- L Legal consultation Juvenile Justice: Who Needs It?...
- F Food and Cooking What age is appropriate for giving your child cocoa?...
Answers on questions: Chemistry
- C Chemistry Ejemplos de moleculas quirales...
- C Chemistry While looking at calcium (Ca) on the periodic table, a student needs to find an element with a greater atomic mass in the same period. Where should the student look? directly up directly...
- C Chemistry Plz finish question asap worth 25 pts the is found in the top part of the and it absorbs much of the suns uv radiation...
- C Chemistry Which of the following does not change the energy of the reactants relative to the activation energy? reducing the temperature adding a catalyst increasing the surface area increasing...
- C Chemistry Write a complete balanced word equation for the following synthesis reaction; sodium metal is added to chlorine gas to produce sodium chloride....
- C Chemistry Any guy wanna be my best friend...
- C Chemistry One kilogram of water (v= 1,003 cm /kg) in a piston/cylinder device at 25°c and 1 bar is compressed in a mechanically reversible, isothermal process to 1,500 bar. determine the amount...
- C Chemistry Boiling water at 1 bar may require a surface heat flux of 97.7 kw m 2 for a surface temperature of 110 °c. what is the value of the heat transfer coefficient?...
- C Chemistry Agas mixture consists of 7 lbm of o2, 8 lbm of n2, and 10 lbm of co2. determine (a) the mass fraction of each component, (b) the mole fraction of each component, and (c) the average...
- C Chemistry Decomposition of radium. radium decomposes at a rate proportional to the quan- if at the end of 20 years approximatelr tity present. 1% of the original quantity has decomposed, how...

Ответ:
142.0 moles CO₂
Explanation:
To answer this question, we first need to know what a mole is. A mole represents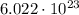 of something, and in this case, that something is molecules.
of something, and in this case, that something is molecules.
So, to convert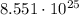 molecules into moles, we need to use the conversion factor
molecules into moles, we need to use the conversion factor 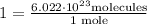 .
.
Doing so (using dimensional analysis) gives us: