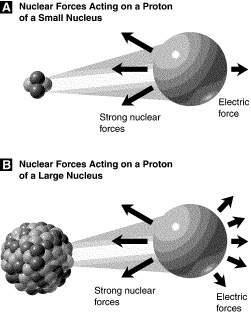
Study figure 10-1. the strong nuclear force felt by a single proton in a large nucleus
a. is much greater than that felt by a single proton in a small nucleus.
b. is much less than that felt by a single proton in a small nucleus.
c. is about the same as that felt by a single proton in a small nucleus.
d. is about the same as the electric force felt by a single proton in a small nucleus.
Solved
Show answers
More tips
- S Science and Technology How to Restore the Diamond Shine to Your Tarnished Silverware...
- C Computers and Internet Boost your processor performance with these easy tips...
- L Leisure and Entertainment When will Maslenitsa start?...
- F Food and Cooking Discovering the Mysterious Fruit of Feijoa...
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- W Work and Career How to Write a Resume That Catches the Employer s Attention?...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...

Ответ:
Explanation:
The strong nuclear force is one of the four forces in nature: 1) strong nuclear force, 2) weak nuclear force, 3) electrostatic force, and 4) gravity.
Strong nuclear force is the strongest of the forces and is resposible for the attraction of the protons and neutrons in the nucleus, and so it is the responsibile for the stability of the atom nuclei.
Neverthelss, it is also a force of very low range it can act only at very small distances. That means that the larger the nucleus the weaker the force.
That is the reason for the unstability of the large atoms.
Ответ:
3.7 L
Explanation:
Initial volume (V₁) = 3.3 L
Initial pressure (P₁) = 100 KPa
Final pressure (P₂) = 90 KPa
Final volume (V₂) =?
The final volume of the gas can be obtained by using the Boyle's law equation as follow:
P₁V₁ = P₂V₂
100 × 3.3 = 90 × V₂
330 = 90 × V₂
Divide both side by 90
V₂ = 330 / 90
V₂ = 3.7 L
Thus, the final volume of the gas is 3.7 L