SupremeNerdx
08.04.2020 •
Chemistry
. Sulfur dioxide can be produced in the laboratory by the reaction of hydrochloric acid and a sulfite salt such as sodium sulfite. Na2SO3 2HCl £ 2NaCl SO2 H2O What mass of SO2 can be made from 25.0 g of Na2SO3 and 22.0 g of HCl?
Solved
Show answers
More tips
- H Health and Medicine How to Treat Whooping Cough in Children?...
- L Leisure and Entertainment Unlocking the Secrets of Fast and Effective Tectonic Learning...
- A Animals and plants Уход за джунгариками: полезные советы и рекомендации...
- S Style and Beauty How to knit a hooded cowl?...
- S Style and Beauty How to Break in New Shoes: 7 Simple Methods...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- A Art and Culture How to Learn Screaming: Step-by-Step Guide for Beginners...
- A Art and Culture Attention, the Final Episode of Margo is Almost Here!...
- H Health and Medicine Novomin: What is it and how to use it?...
- L Leisure and Entertainment How to Land on the Moon: Your Comprehensive Guide...
Answers on questions: Chemistry
- C Chemistry Calculate the concentration in molality of calcium phosphate in a solution containing 250.0 grams of pure water when the freezing point is -8.5 c...
- C Chemistry A3.87mg sample of an organic compound gave 5.80mg co² and 1.58mg h²o on combustion. deduce the empirical formula of the compound....
- C Chemistry Is helium more reactive than magnesium...
- C Chemistry Dhbwjkdnjhbnjfhgdvbnjfuyhgevbdnjfuhgbvf...
- C Chemistry A compound is composed of 4.80 g of carbon and 0.400 g of hydrogen. The molar mass of the compound is 78.0 g....
- C Chemistry An object has a mass of 20 g and volume of 10 cm3. Enter the numerical value for the density of the object in units of g/cm3....
- C Chemistry All atoms of the same elements have the same what?...
- C Chemistry Could a juman stay alive if one body system is not working? could the body maintain homeostasis?...
- C Chemistry Pls help its due tomorrow, just help with as many questions as you can, I need to show work using the factor label method....
- C Chemistry In an ionic bond, select one: a. atoms, having gained or lost electrons, attract one another with opposite charges. b. two atoms are attracted by the same charges. c. atoms...

Ответ:
Mass of SO₂ can be made from 25.0 g of Na₂SO₃ and 22 g of HCl = 12.672 g
Explanation:
SO₂( sulfur dioxide) can be produced in the lab. by the reaction of hydrochloric acid & sulphite salt such as sodium.
the balanced chemical equation is as follows
Na₂SO₃ + 2 HCl → 2 NaCl + SO₂ + H₂O
Moles of Na₂SO₃ =
Moles of HCl =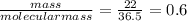
using mole ratio method to find limiting reagent
For sodium sulfite
for HCl
since sodium sulfite is limiting reactant for above chemical reaction
1 mole of Na₂SO₃ produce 1 mole of SO₂
0.198 mole of Na₂SO₃ produce 0.198 mole of SO₂
∴ Mass of SO₂ produce = mole x molar mass of SO₂
= 0.198 x 64
= 12.672 g
Ответ:
im not certain but i think C and then A
Explanation: