elielson1497
25.11.2021 •
Chemistry
the absence of c5 means that the molecule is not present to be activated. what happens in normal serum when c5 is ""activated""?
Solved
Show answers
More tips
- H Health and Medicine How Did Inna Lose Weight on Dom 2?...
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
Answers on questions: Chemistry
- C Chemistry Fast : is this right.. if not.. which one is it? fast : (...
- C Chemistry 76/304=81/k what is k and show work plez...
- C Chemistry Flour is a white solid that burns and does not dissolve in water. Sugar is a white solid that dissolves in water and will turn brown and give off a sweet odor when...
- C Chemistry What is the mass of 2.5 mol of ca, which has a molar mass of g/mol...
- H History Eritrea is a landlocked nation truefalse...
- M Mathematics A set of three scores consists of the values 6, 3, and 2. Σ2(X – 3)...
- M Mathematics Find a, geometry help !...
- C Chemistry Es un 1 punto¿Cuál de los siguientesmaterial conductor deelectricidad? *O ÁrbolO LlaveO Hoja...
- E English Question 9 of 10 Which development would most likely reduce the standard of living in the United States? O A. Wages increase for most American workers. O B. It...
- M Mathematics A city receives 3 inches of snowfall every 6 hours. Write and graph a function that describes the relationship. How long does it take to receive 1 foot of snow?...

Ответ:
Further Explanation:
Ideal gas:
An ideal gas contains a large number of randomly moving particles that are supposed to have perfectly elastic collisions among themselves. It is just a theoretical concept, and practically no such gas exists. But gases tend to behave almost ideally at a higher temperature and lower pressure.
Ideal gas law is considered as the equation of state for any hypothetical gas. The expression for the ideal gas equation of gas is as follows:
Here,
P is the pressure of the gas.
V is the volume of gas.
n denotes the number of moles of gas.
R is the gas constant.
T is the temperature of gas.
Boyle’s law:
It is an experimental gas law that describes the relationship between pressure and volume of the gas. According to Boyle's law, the volume of the gas is inversely proportional to the pressure of the system, provided that the temperature and the number of moles of gas remain constant.
If the temperature and number of moles of gas are constant then the equation (1) will become as follows:
Or it can also be expressed as follows:
Here,
Rearrange the equation (3) for , and we get,
, and we get,
The conversion factor to convert into
into 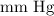 is as follows:
is as follows:
The value of is 2.50 atm.
is 2.50 atm.
The value of is 25 mL.
is 25 mL.
The value of is 457 mm Hg.
is 457 mm Hg.
Substitute these values in equation (4).
The final volume of a gas is 103.94 mL.
Learn more:
1. Determine the final pressure of the ideal gas:
2. Chemical bonds in NaCl:
Answer details:
Grade: Senior School
Subject: Chemistry
Chapter: Ideal gas equation
Keywords: ideal gas, pressure, volume, absolute temperature, equation of state, hypothetical, universal gas constant, moles of gas, initial, final.