tatianna341
06.07.2019 •
Chemistry
The air inside a bubble gum bubble is at atmospheric pressure. imagine someone continuing to blow into that bubble. what happens when the bubble’s volume is doubled? a. the number of air particles in the bubble is cut in half. b. the number of air particles in the bubble is doubled. c. the bubble’s temperature is cut in half. d. the bubble’s temperature is doubled.
Solved
Show answers
More tips
- F Food and Cooking Learn How to Make Ice Cream at Home - Step by Step Guide...
- H Health and Medicine When can it be said that a person has a normal pulse?...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- F Food and Cooking How to Properly Cook Buckwheat?...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
Answers on questions: Chemistry
- C Chemistry When a bat hits a ball, what is the impulse? the force multiplied by the time the objects are in contact the mass multiplied by the acceleration the change in velocity...
- C Chemistry Most ionic compouds are crystalline solids at room temperature. true falseionic compounds are electrically neutral. true falseionic compounds generally have low melting...
- C Chemistry Which of the following pairs of elements is most likely to form an ionic compound? a oxygen and fluorine b sodium and aluminum c calcium and chlorine d nitrogen and...
- C Chemistry In the reaction below, if 5.71 g of sulfur is reacted with 10.0 g of oxygen, how many grams of sulfur trioxide will be produced?...
- C Chemistry Drag each label to the correct image. each label can be used more than once. label the samples based on their ph values. alkaline acidic neutral lemon juice ph 2.2 salt...
- C Chemistry determine the empirical formula of a compound containing 48.38 G of carbon 26.74 grams of hydrogen and 53.5 grams of oxygen in a experiment the molar mass of the compound...
- C Chemistry Which expression represents the concentration of OH– in solution? a. 10–14 / [H3O+] b. [OH–] / 10–14 c. 10–14 – [H3O+] d. 10–14 x [H3O+]...
- C Chemistry Consider the following balanced equation. 2Al+3H2SO4⟶Al2(SO4)3+3H22Al+3H2SO4⟶Al2(SO4)3+3H2 How many grams of aluminum sulfate will be produced from 28.5 grams of Aluminum?...
- C Chemistry all the stars we observe from earth emit light that is shifted, to varying degrees, to longer wavelengths. This evidence is used to support the big bang theory because...
- M Mathematics Ricardo is building a new garden and is buying soil to fill it up. he buys 4 kilograms of soil for $10.00. what is the unit rate for the soil in kilograms?...

Ответ:
The correct answer is option D. The bubble’s temperature is doubled.
According to Gay-Lussac's law or the pressure law at constant volume of an ideal gas, the pressure exerted is directly proportional to its absolute temperature.
As more air is blown into the bubble the pressure increases which cause the temperature of the air inside the bubble to rise.
Ответ:
Option (D) is the correct answer.
Explanation:
According to Charles' law, at constant pressure volume of an ideal gas is directly proportional to the absolute temperature.
Mathematically, V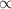 T at constant P
T at constant P
Therefore, when volume of a bubble gum is doubled by blowing air inside it then there will also be two times increase in its temperature since volume is proportional to temperature.
Therefore, we can conclude that when the bubble’s volume is doubled then the bubble’s temperature will also get doubled.
Ответ: