The bonds in the reactants of figure 7-3 contained 372 kj of chemical energy and the bonds in the products contained 350 kj of chemical energy. what is the amount of energy change during the reaction (show your work for full credit)? would this energy be absorbed or released? explain how you know.

Solved
Show answers
More tips
- F Family and Home How to Remove Tar Stains: Tips and Recommendations from Experts...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- S Sport How to Do a Jumping Split...
- H Health and Medicine How Did Inna Lose Weight on Dom 2?...
- F Family and Home How to Properly Fold Napkins in a Napkin Holder?...
- F Food and Cooking How to Set Up Ventrilo - The Ultimate Guide...
- S Science and Technology How to Make a Homemade Smoker: The Ultimate Guide...
- A Auto and Moto Battle for the Relocation of The Cherkizovsky Market: Who Won?...
- C Computers and Internet How Do You Refill Cartridges?...
- S Style and Beauty Tricks and Tips: How to Get Rid of Freckles...
Answers on questions: Chemistry
- M Mathematics Help me please my math didn’t teach me this!...
- F French Assign the correct gender and number....
- C Chemistry PLEASE HELPPP please explain the formula for heat transfer please like what the stuff stands for...
- M Mathematics Leo bought 4 T-shirts and 2 pairs of pants for Php 3 000.00. If the T- shirts cost Php 300.00 each, find the cost of each pair of pants.1) What is asked?2) What are the given?3) What...

Ответ:
22 kJ amount of energy is released in the following reaction.
Explanation: There are two types of reaction on the basis of amount of heat absorbed or released.
1. Endothermic reactions: These are the type of reactions in which reactants absorb heat to form the products. The energy of the reactants is less than the energy of the products.
2. Exothermic reactions: These are the type of reactions in which heat is released from the chemical reactions. The energy of the products is less than the reactants.
Sign convention for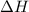 : This value is negative for exothermic reactions and positive for endothermic reactions.
: This value is negative for exothermic reactions and positive for endothermic reactions.
For the given chemical reaction,
Energy of the products is less than the energy of the reactants, Hence, this reaction will be a type of exothermic reaction and energy will be released during this chemical change.
Amount of energy released = (350 - 372) kJ = -22kJ
Negative sign symbolizes the energy is being released. So, 22 kJ amount of energy is released in the following reaction.
Ответ:
Explanation:
1. Increasing the concentrations of reactants in a chemical reaction would affect a reaction positively by increasing the number of collisions within a particular period which in turn increases the rate of reaction.
2. The molecules gains energy,which enable them to move faster and collide with the right energy for a reaction to take place.
3. The average kinetic motion of molecules in a solution can be measured by measuring the temperature. Temperature is a measure of the average kinetic energy of molecules in a solution.