alannaamarriee
10.03.2020 •
Chemistry
The chemical equation shows iron(III) phosphate reacting with sodium sulfate. 2FePO4 + 3Na2SO4 Fe2(SO4)3 + 2Na3PO4 What is the theoretical yield of Fe2(SO4)3 if 20.00 g of FePO4 reacts with an excess of Na2SO4
Solved
Show answers
More tips
- H Health and Medicine Headache: A Comprehensive Guide to Treatment...
- G Goods and services Stock center - a modern way of optimizing logistics...
- F Food and Cooking How to Properly Wash a Down Jacket? Tips from Experts...
- C Computers and Internet Thin Client: What It Is and Why You Need It?...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
Answers on questions: Chemistry
- C Chemistry * 4. Is the moon a planet? O yes O no O maybe...
- C Chemistry What property of water makes it a good choice to use to cool down hot material quickly ?...
- C Chemistry Put all chemicals down the drain. true or false?...
- M Mathematics Reflect triangle A in the line y = 1....
- H History Which of the following is an impact of the lack of uniformity in the international banking system? O A. Investors are uninformed. O B. Investors can make large profits....
- B Business XYZ stock is trading at $25.75 and XYZ Jul 25 calls are trading at a premium of $2. What is the time value of the Jul 25 calls...
- P Physics A 1.97-pF capacitor is connected to a 9.0-V battery and fully charged. How many electrons did the battery transfer from one capacitor plate to the other...
- M Mathematics The Insurance Institute for Highway Safety publishes data on the total damage suffered by compact automobiles in a series of controlled, low-speed collisions. A sample...
- E English “A child longs for toys, sweets and other things, but not at the cost of his parents”. Comment from The lost child...
- M Mathematics 1. Complete the table below to solve the equation 2.5x − 10.5 = 64(0.5 ). 2. A newspaper started an online version of its paper 14 years ago. In a recent presentation...

Ответ:
The theoretical yield of iron(III) sulfate is 26.6 grams
Explanation:
To calculate the number of moles, we use the equation:
Given mass of iron(III) phosphate = 20.00 g
Molar mass of iron(III) phosphate = 150.82 g/mol
Putting values in equation 1, we get:
The given chemical equation follows:
As, sodium sulfate is present in excess. So, it is considered as an excess reagent.
Thus, iron(III) phosphate is considered as a limiting reagent because it limits the formation of product.
By Stoichiometry of the reaction:
2 moles of iron(III) phosphate produces 1 mole of iron(III) sulfate
So, 0.133 moles of iron(III) phosphate will produce =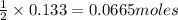 of iron(III) sulfate
of iron(III) sulfate
Now, calculating the mass of iron(III) sulfate from equation 1, we get:
Molar mass of iron(III) sulfate = 399.9 g/mol
Moles of iron(III) sulfate = 0.0665 moles
Putting values in equation 1, we get:
Hence, the theoretical yield of iron(III) sulfate is 26.6 grams
Ответ:
I think it’s no tbh