Demaryaflie
06.05.2020 •
Chemistry
The contents of a rock have a 206Pb to 238U mass ratio of 0.135:1.00. Assuming that the rock did not contain any 206Pb at the time of its formation, determine the age of the rock. Uranium-238 decays to lead-206 with a half-life of 4.5 billion years. Express the time to two significant digits.
Solved
Show answers
More tips
- S Science and Technology When do we change our clocks?...
- A Animals and plants How to Teach Your Parrot to Talk?...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- F Food and Cooking How to Make Polendwitsa at Home?...
- F Family and Home Parents or Environment: Who Has the Most Influence on a Child s Upbringing?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- C Chemistry If you gently shake a carbon di oxide fire extinguisher you will feel the presence of liquid within the extinguisher.what condition shoud be maintained so that carbon...
- C Chemistry To turn water into ice do you need to add or to subtract kinetic energy?...
- C Chemistry The stick bug shown in the picture below evades predators by blending in with its surroundings. What type of adaptation does this represent? a mimicry b. camouflage...
- C Chemistry Calculate the work (kJ) done during a reaction in which the internal volume expands from 30 L to 60 L against a vacuum (an outside pressure of 0 atm)....
- C Chemistry Acarbon atom is most likely to form what kind of bond(s) with other atoms? ionic bonds, covalent bonds, and hydrogen bonds covalent ionic hydrogen covalent bonds...
- C Chemistry The different forms of a gene for a given trait are called?...
- H History Was Comte correct in believing that once identified the laws that governed society, sociologists could address problems such as poor education and poverty? Why or...
- B Biology Your task As chief investigators of the South Carolina law enforcement division, it is your job to analyze the data to determine who stole the money and what their...
- M Mathematics Danny spent $37.35 on 15 gallons of gasoline. What was the price per gallon?...
- H History For what reason did the second Texas president, Mairabeau B. Lamar move the Texas capital from Houston to Austin...

Ответ:
The age of the rock is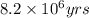
Explanation:
The equation used to calculate rate constant from given half life for first order kinetics:
where,
Putting values in above equation, we get:
We are given:
Mass ratio of 206-Pb to 238-U = 0.135 : 1.00
Rate law expression for first order kinetics is given by the equation:
where,
k = rate constant =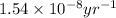
t = time taken for decay process = ? yr
[A] = amount left after decay process = 1.00 grams
Putting values in above equation, we get:
Hence, the age of the rock is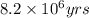
Ответ:
2) Chloroform & Caustic potash
Explanation:
The carbylamine reaction is a kind of chemical test which is done to detect primary amines in an unknown solution. It cannot detect secondary and tertiary amines.
The reaction involves the heating with up of the unknown solution with alcoholic potassium hydroxide or caustic potash and the chloroform.
In the presence of primary amine, the production of isocyanide results.