The degree of ionization of a weak acid I. varies with the concentration of the acid. II. depends on which weak acid it is. III. is 100%. IV. is greater than 50% but less than 100%.
Solved
Show answers
More tips
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- D Dating, Love, Relationships How to Overcome Jealousy: Tips and Tricks...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- C Computers and Internet How to Learn to Type Fast?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
Answers on questions: Chemistry
- C Chemistry What is Atomic Radius...
- C Chemistry Hope this helps with my last question or I wasted my points...
- C Chemistry Which element below is the most reactive? oxygen hydrogen chlorine neon...
- C Chemistry Where exactly in our solar system is this water found?...
- C Chemistry Write the equilibrium expression, calculate keq and then tell where the equilibrium lies: fe (s) + o2 (g) ↔ fe2o3 (s) in a 2.0 l container at equilibrium: fe = 1.0 mol o2 = 1.0 e-3...
- A Arts Who wants to join a roleplay padlet? :D...
- S Social Studies A person you admire most...
- M Mathematics Evaluate the function f(x) = 2x + 8, when x = 0...
- H History Was the Amircan revolution on France?...
- P Physics If a dog runs 25m east and then turns around and runs back 20 west, what is the total distance ran? How far is the dog displacement from the starting line?...

Ответ:
The degree of ionization of a weak acid is greater than 50% but less than 100%.
Explanation:
An acid which dissociates partially or weakly when dissolved in water is called a weak acid.
For example, acetic acid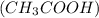 is a weak acid.
is a weak acid.
Hence, the degree of ionization of a weak acid is greater than 50% but less than 100%.
Thus, we can conclude that the degree of ionization of a weak acid is greater than 50% but less than 100%.
Ответ:
Its 2 man
Explanation: