ayoismeisalex
15.01.2020 •
Chemistry
The diameter of the tungsten wire in a lightbulb filament is very small, less than two thousandths of an inch, or about 1/20 mm. the mass of the filament is so very small - 0.0176 grams - that it would take 1600 filaments to weigh an ounce! how many tungsten atoms are in a typical lightbulb filament?
Solved
Show answers
More tips
- G Goods and services How to Make Soap at Home: Useful Tips for Beginners...
- C Computers and Internet How to Reinstall Windows: A Detailed Guide for Beginners...
- S Style and Beauty How to Get Rid of a Bruise: Tips and Tricks...
- F Food and Cooking Лечо: вкусное и простое блюдо для любой кухни...
- H Health and Medicine Relieving Swelling in Legs: Causes and Ways to Alleviate the Symptom...
Answers on questions: Chemistry
- C Chemistry The law of conservation of mass states that matter can neither be created on destroyed. This is shown when a chemical reaction has an number of atoms on both sides...
- C Chemistry Why is wood harder to use as fuel than oil?...
- C Chemistry Calculate the molar mass of mgoh2...
- C Chemistry Juhygg ggvjjh gdhdhdhhdhdh...
- C Chemistry PLs help pls does anyone know this pls...
- C Chemistry How might you modify the popcorn experiment to find out whether the presence of duds did or did not make a difference in my calculations...
- C Chemistry Why can iron be broken down into atoms, but water can only be broken down to molecules?...
- C Chemistry Will a tower of straws hold tennis balls...
- C Chemistry Please send me your what.sapp number ...
- C Chemistry In an expenmerk wooom of 0.119Mbanum hydroxide were mixed with 200 mg 0.330M aluminium sulphate. what is(total massof theme breubitak test forhuc3...

Ответ:
A typical lightbulb filament, whose mass is 0.0176 g, contains 5.76 × 10¹⁹ atoms of tungsten.
The mass of a typical lightbulb filament of tungsten is 0.0176 grams. We can convert 0.0176 g to moles using the molar mass of tungsten (183.84 g/mol).
To convert 9.57 × 10⁻⁵ mol to atoms, we will use Avogadro's number: there are 6.02 × 10²³ atoms in 1 mole of atoms.
A typical lightbulb filament, whose mass is 0.0176 g, contains 5.76 × 10¹⁹ atoms of tungsten.
Learn more: link
Ответ:
The number of tungsten Atoms in a typical light bulb filament is
5.76 ×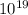 atoms of W
atoms of W
Explanation:
Write the given values of the problem
The diameter the tungsten wire in a light bulb filament Tungsten element W : molar mass = 183.84 g / mol
0.0176 g W ×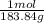 = 0.000096 mol W
= 0.000096 mol W
0.000096 mol W × }{1 mol W}" alt="10^{23 atoms}" />}{1 mol W}" />
}{1 mol W}" alt="10^{23 atoms}" />}{1 mol W}" />
= 5.76 ×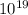 atoms of W
atoms of W
Ответ:
true
Explanation: