lightning1157blaze
06.04.2020 •
Chemistry
The energy given off by electrons is referred to as .
Solved
Show answers
More tips
- F Food and Cooking Delicious and Simple Fillings for Pies...
- H Health and Medicine How to Help Men Gain Weight?...
- A Auto and Moto Discovering the Leader: What is the Most Expensive Car in the World?...
- H Health and Medicine Hangover: How to Get Rid of It Quickly?...
- S Style and Beauty How to Choose the Right Fur Coat and Avoid Regrets?...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- M Mathematics In inverse variation, the variables change by a constant...
- S Social Studies Why is your Catalog year important? A. It corresponds to the Degree Requirements required for you Everyone is always in the most recent catalog year B. It is the year you will...
- H History The kiwi is a flightless bird that lives in what country?...

Ответ:
When the electron returns to its original position, an electromagnetic wave is produced. Depending on the kind of atom and the amount of energy, this electromagnetic radiation can take the form of heat, light, ultraviolet, or other electromagnetic waves. There are several ways of causing atoms to absorb energy.
Explanation...
Ответ:
1)Thus based on the diagram , the values of principle quantum number that can be assigned to the electrons are : 2,3 and 4.
2)This quantum number tells us the shell in which an electron is present and how far it is roughly from the nucleus.
Explanation:
According to the rules of electron filling in subshells in quantum mechanics , Aufbau's principle states that :
"An electron must be filled in a lower energy state or subshell and then higher. If they have the same energy , the one with lesser principal quantum number will be filled first."
Thus ,
The series of filling goes like:
Thus , the subshells in the given information have 1,3 and 1 orbitals respectively. Thus it has to be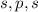 which is possible in two cases :
which is possible in two cases :
1)Thus based on the diagram , the values of principle quantum number that can be assigned to the electrons are : 2,3 and 4.
2)This quantum number tells us the shell in which an electron is present and how far it is roughly from the nucleus.