pstrezze1840
30.11.2019 •
Chemistry
The flavor of anise is due to anethole, a compound with the molecular forumal c10h12o. combustion of one mole of anethole produces 5541 kj of thermal energy. if 0.950 g of anethole is combusted in a bomb calorimeter whose heat capacity (ccalorimeter) is 7.854 kj/°c, what is the change in temperature of the calorimeter?
Solved
Show answers
More tips
Answers on questions: Chemistry
- C Chemistry Molecules containing two carbon atoms and a functional group have many home and industrial uses. these compounds can be produced by a variety of reactions, as shown...
- C Chemistry The value of(2.3) - 0.027(2.3)2 +0.69 +0.09is :...
- C Chemistry If the amount of radioactive chromium-51, used to label red blood cells, in a sample decreases from 1.6 mg to 0.40 mg in 55.4 days, what is the half-life of chromium-51?...
- C Chemistry Why do most people think that I am a guy on this app?...
- C Chemistry What conducts better, metal or nometal? why?...
- C Chemistry Given 57 grams of NO2, A. How many moles of NO2, are present? B. How many atoms of oxygen are present?...
- C Chemistry If the [Hg2 +] in a 0.10 M solution of the Hg (Cl4)^2- complex is 1.30 x 10^-4 M, calculate the dissociation constant for the complex....
- M Mathematics Find the value of the missing angle....
- S Social Studies Forgetting takes place only in short-term memory. Please select the best answer from the choices provided T F....
- B Business During periods of inflation, which inventory method will typically yield the highest cost of goods sold...

Ответ:
Change in temperature of calorimeter is
Explanation:
Molar mass of anethole = 148.2 g/mol
So, 0.950 g of anethole =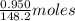 of anethole = 0.00641 moles of anethole
of anethole = 0.00641 moles of anethole
1 mol of anethole releases 5541 kJ of heat upon combustion
So, 0.00641 moles of anethole release of heat or 35.52 kJ of heat
of heat or 35.52 kJ of heat
7.854 kJ of heat increases temperature of calorimeter.
temperature of calorimeter.
So, 35.52 kJ of heat increases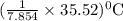 or
or  temperature of calorimeter
temperature of calorimeter
So, change in temperature of calorimeter is
Ответ:
They basically use them to summarize what happens in chemical reactions
Explanation:
hope this helps :)