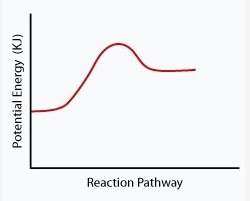
The following phase diagram shows how a catalyst affected the rate of a reaction.
which statement below best describes how the catalyst affected the reaction shown in this diagram?
a. the catalyst decreased the activation energy of the reactants.
b. the catalyst increased the activation energy of the reactants.
c. the catalyst increased activation energy of the products.
d. the catalyst decreased activation energy of the products.
Solved
Show answers
More tips
- P Philosophy Why Did God Create Man and Place Him in Obscurity?...
- S Science and Technology How to Make a Homemade Smoker: The Ultimate Guide...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry Which processes explain how cells with the same genetic information can produce cells with unique structures and therefore functions? Select all that apply. a. Mitosis b...
- C Chemistry The production of liquid ammonia (NH3) comes from the combination of Nitrogen Gas (N2) and Hydrogen Gas (H2). If you had a tanker (5000.0 L) of hydrogen gas at 100.0 K and...
- C Chemistry Can someone help me plz I’m struggling...
- C Chemistry Qual a razão que encontras, para a dureza da água se incluir no estudo das reações químicas?...
- C Chemistry Name two methods that can be used to break down compounds into simpler substance...
- C Chemistry What family or group is neon in? like a halogen, alkali metal or something. (im doing a chemistry worksheet on neon and i suck at chemistry.)...
- C Chemistry The thin outside layer of the Earth is called the crust mantle core O dirt...
- C Chemistry A radioisotope of the element carbon (10C) decays to an isotope of the element boron by emission of a positron. Complete the equation for the decay process....
- C Chemistry Certain reactions in cells are endergonic. how do cells make those reactions proceed?...
- C Chemistry If two sulfur atoms wanted to bond together, how many electrons would each sulfur share with the other to reach their desired total of 8?...

Ответ:
The correct answer is "The catalyst decreased the activation energy of the reactants."
Explanation:
Activation energy is the minimum energy required to start a reaction. A catalyst lowers the activation energy of a reaction. Here the catalyst lowers the activation energy of the reactant, thus making it easier to start to the reaction. In the absence of a catalyst the activation energy of the reactant will be higher.
Ответ:
"electric potential difference is the difference in electric potential (V) between the final and the initial location when work is done upon a charge to change its potential energy. In equation form, the electric potential difference is.."
check the link below. very helpful!
Explanation:
https://www.physicsclassroom.com/class/circuits/Lesson-1/Electric-Potential-Difference