The following reaction proceeds at a rate such that 3 mole of a is consumed per minute. given this, how many moles of c are produced per minute? 2a+2b→4c express your answer with the appropriate units.
Solved
Show answers
More tips
- C Computers and Internet How Much Does an iPhone Cost in America?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- H Health and Medicine Headache: A Comprehensive Guide to Treatment...
- C Computers and Internet How to Properly Order Clothing from International Online Stores...
- A Auto and Moto Mastering One-Movement Parking: All You Need to Know...
- D Dating, Love, Relationships How Long Can Love Last?...
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- F Food and Cooking How to Find Your Zip Code?...
- S Style and Beauty How to choose the best mascara for your eyelashes...
- S Style and Beauty How to Apply Foundation Correctly?...
Answers on questions: Chemistry
- C Chemistry An indicator of average kinetic energy is temperature pressure volume diffusion...
- C Chemistry If kerosene has a specific gravity of 0.820, what force will be exerted on the circular bottom of a cylindrical kerosene tank that has a diameter of 12-ft and a height of 30 ft?...
- C Chemistry The combustion of 135 mg of a hydrocarbon produces 440 mg of CO2 and 135 mg H2O. The molar mass of the hydrocarbon is 270 g/mol. Determine the molecular formula of this compound....
- C Chemistry Which can change from one state of matter to another at the temp nitrogen water oxygen carbon dioxide...
- C Chemistry Which material best absorbs sound waves in a room? a. heavy curtains b. hardwood floors c. brick walls d. cement floors...
- C Chemistry A certain gas is present in a 10.0 LL cylinder at 4.0 atmatm pressure. If the pressure is increased to 8.0 atmatm the volume of the gas decreases to 5.0 LL . Find the two constants...
- C Chemistry What would be the BMI for a person who is 188 cm and weighs 90.7 kg? For a person who is 163 cm and weighs 108.9 kg?...
- C Chemistry If the reaction of 91.3 grams of C3H6 produces a 81.3% yield, how many grams of CO2 would be produced?...
- C Chemistry If you reacted 10 moles of Propane with 10 moles of Oxygen, how many moles of propane would you consume? How many moles of carbon dioxide would you generate? How much carbon dioxide...
- C Chemistry State the difference between Allotropy and Polymorphism...

Ответ:
6 moles of C are produced per minute
Explanation:
According to law of mass action for the given reaction-
Rate =![-\frac{1}{2}\frac{\Delta [A]}{\Delta t}=-\frac{1}{2}\frac{\Delta [B]}{\Delta t}=\frac{1}{4}\frac{\Delta [c]}{\Delta t}](/tpl/images/0224/0618/86af6.png)
Where![-\frac{\Delta [A]}{\Delta t}](/tpl/images/0224/0618/185ce.png) represents rate of consumption of A and
represents rate of consumption of A and ![\frac{\Delta [C]}{\Delta t}](/tpl/images/0224/0618/e549e.png) represents rate of production of C
represents rate of production of C
Here![-\frac{\Delta [A]}{\Delta t}](/tpl/images/0224/0618/185ce.png) = 3 moles/min
= 3 moles/min
So,![\frac{\Delta [C]}{\Delta t}](/tpl/images/0224/0618/e549e.png) =
= ![\frac{4}{2}\times -\frac{\Delta [A]}{\Delta t}](/tpl/images/0224/0618/941aa.png)
So,![\frac{\Delta [C]}{\Delta t}](/tpl/images/0224/0618/e549e.png) =
= 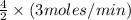
So,![\frac{\Delta [C]}{\Delta t}](/tpl/images/0224/0618/e549e.png) = 6 moles/min
= 6 moles/min
Ответ: