The half-life of the radioactive element unobtanium dash 53 is 20 seconds. if 32 grams of unobtanium dash 53 are initially present, how many grams are present after 20 seconds? 40 seconds? 60 seconds? 80 seconds? 100 seconds?
Solved
Show answers
More tips
- O Other What is the oldest joke ever told?...
- F Food and Cooking How to Make Lazy Cabbage Rolls? Simple Steps to a Delicious Dish...
- F Food and Cooking Unusually Delicious Shashlik - Follow the Etiquette of Proper Preparation!...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- G Goods and services Which TV is better - LCD or Plasma?...
- S Sport How to Learn to Pull Up on Monkey Bars?...
- L Leisure and Entertainment Scrapbooking: What is it and Why is it Becoming More Popular?...
- C Computers and Internet Where did torrents.ru move to?...
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
Answers on questions: Chemistry
- C Chemistry I need an answer please I’m desperate...
- C Chemistry The Lithium atom has only two energy levels. The third is empty. Therefore, we can refer to the second energy level of the lithium atom as the outermost level. The electrons...
- C Chemistry increasing the volume of a given amount of gas at constant temperature cause the pressure to decrease because...
- C Chemistry A student used 16.5 cm3 of nitric acid to neutralise 25 cm³ of 0.4 mol dm-3 sodium hydroxide solution. What is the concentration of nitric acid used? A)0.61mol dm-3B)0.40mol...
- C Chemistry In a chemical formula, the letters tell you the?...
- E English You have been installed a chief in your hometown, Write a litter to your friend in another school telling him at least two things you intend doing to develop the town....
- M Mathematics 1 A tree in Keiko s backyard has been growing for 5 2 years. The tree is 12 / 2 meters tall. What is the unit rate in meters per year? Write your answer as a fraction or a...
- M Mathematics Number 17 !! tyy, im rlly bad at math ong...
- M Mathematics I Need help with number 9...
- M Mathematics What is x?: 8+9.8x+9=3.8x - 16+3.8x...

Ответ:
The half-life of a radioactive element is defined as the time required by the specific isotope to decrease by half of its original value. The unobtanium after given time will be present as:
After 20 secs, 16 gramsAfter 40 secs, 8 gramsAfter 60 secs, 4 gramsAfter 80 secs, 2 gramsAfter 100 secs, 1 gramHalf-life is the time required by the unobtanium is the half of the reaction is completed. Half of the substance will be decomposed, such that:
Amount of substance left n half-lives =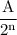
where A= initial amount of substance
Now,
n = number of half-lives = given time /half-life
Given,
1. Time = t = 20 seconds
Number of half-lives =2.Time = t = 40 seconds
Number of half-lives =3.Time = t = 60 seconds
Number of half-lives =4.Time = t = 80 seconds
Number of half-lives =5.Time = t = 100 seconds
Number of half-lives =To know more about half-life, refer to the following link:
link
Ответ:
After 20 seconds, 16 grams of unobtanium will be present, after 40 seconds, 8 grams of unobtanium will be present after 60 seconds, 4 grams of unobtanium will be present, after 80 seconds, 2 grams of unobtanium will be present and after 100 seconds, 1 grams of unobtanium will be present.
Explanation: Half life is the time in which half of the reaction is completed. Thus the half of the substance will be decomposed and half of it will remain.
Amount of the substance left after n half lives will be=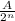
where A= initial amount of substance
n=no of half lives=
a) t= 20 seconds
no of half lives=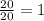
amount of the substance left after 1 half life will be= =16 g.
=16 g.
b) t= 40 seconds
no of half lives=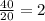
amount of the substance left after 2 half lives will be= =8g.
=8g.
c) t= 60 seconds
no of half lives=
amount of the substance left after 3 half lives will be=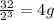
d) t= 80 seconds
no of half lives=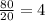
amount of the substance left after 4 half lives will be=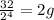
e) t= 100 seconds
no of half lives=
amount of the substance left after 5 half lives will be=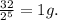
Ответ:
is there more information please