lewismichelle11
03.11.2020 •
Chemistry
The heat of vaporization of a certain refrigerant is 162 J/g. Calculate the number of
kilograms of refrigerant that must be evaporated to freeze a tray of 16 one-ounce
(1 oz = 28 g) ice cubes starting with the water at 16°C.
Heat capacity (water) = 4.184 J/g °C
ΔHfusion (water) = 333 J/g
Solved
Show answers
More tips
- W Work and Career How to Start Your Own Business: Tips and Recommendations...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Computers and Technology Patricia wants to write a program that will determine how many ice cream cones are left over when the scoops of ice cream are divided by the cones. Which operation...
- B Biology I WILL GIVE BRAINLIEST1 Social classes is a characteristic of civilization that refers to groups of people ranked by their place in society people who come together...
- E English Identify the word below that cannot be divided by a hyphen across lines. a. tender b. prefix c. motherland d. idea...
- M Mathematics Some reindeer prefer eating carrots, while others prefer apple. if the ratio of carrot eaters to apple eaters is 30: 420, determine the number of reindeer that...
- M Mathematics To sacrifice my own life for pakistan...

Ответ:
We need 1.106 kilograms of refrigerant to freeze 16 one-ounce ice cubes.
Explanation:
According to the statement, the refrigerant must receive the heat released from water to create ice cubes. If we know that there is no heat nor mass interactions with surroundings and freezing process is steady, then we get the following model from First Law of Thermodynamics:
Where:
Please notice that refrigerant is only vaporized (latent), whereas water is cooled down and freezed (sensible + latent). Then, we expand the equation above by definitions of sensible and latent heat:
Where:
Then we clear the mass of the refrigerant within:
If we know that ,
,  ,
,  ,
, 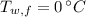 ,
,  and
and  , then the mass of the refrigerant is:
, then the mass of the refrigerant is:
If we know that a kilogram equals 1000 grams, then we need 1.106 kilograms of refrigerant to freeze 16 one-ounce ice cubes.
Ответ:
The first one,The second one, The third one And the forth one
Step-by-step explanation:
I JUST TOLD THE TEST.