The Ksp can be used to find the concentration of hydroxide ions, and thus determine the pH given a concentration of of aluminum. At what pH will precipitation of Al(OH)3 begin if 6.70 lb of aluminum sulfate, Al2(SO4)3, is added to 2050 gallons of water (with a negligible change in volume)?
Solved
Show answers
More tips
- C Computers and Internet How to Learn to Type Fast?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- H Health and Medicine What is Autism? Understanding the Basics of This Neurodevelopmental Disorder...
- P Philosophy How to Develop Extrasensory Abilities?...
- S Style and Beauty Don t Sacrifice Your Brows: How to Properly Pluck Stubborn Hairs...
Answers on questions: Chemistry
- C Chemistry Why is water considered to be neutral?...
- C Chemistry Air is a mixture of gases that is about78.0percent n2 by volume. when air is at standard pressure and 25.0 degreec, the n2 component will dissolve in water with...
- C Chemistry When deployed, car air bags rapidly inflate as sodium azide, NaN₃, decomposes into it s component elements, given by the following reaction: 2 NaN₃- 2 Na + 3 N₂...
- C Chemistry Q. Atmospheric pressure on the peak of Mont Everest can be as low as 0.20 atm. If the volume of an oxygen tank is 10.0L, at what pressure must the tank be filled...
- C Chemistry Im very confused and need help...
- C Chemistry Cierto átomo de número de masa 80 posee una configuración electrónica donde presenta 2 subniveles principales llenos, en base a este dato. Hallar el mínimo valor...
- E English How do we come to understand big concepts ?...
- M Mathematics Two consecutive numbers can be represented by x and x - 1. the product of two consecutive numbers is equal to 35 more than eleven times the sum of the numbers....
- M Mathematics Need on both questions 50 points and branliest...
- M Mathematics Answer the following questions correctly i will know if this is wrong. i will report any incorrect answers! criteria for getting brainliest and not getting reported:...

Ответ:
3.9.
Explanation:
(Molar mass of 342.15 g/mol).
The first thing to do in this question is to coverts our units. We are given 6.70 lb = 453.592 × 6.70 lb = 3039.0664 g of aluminum sulfate.
2050 gallons of water = 2050 × 3.785 = 7759.25 Litres.
After this we will now look for the Concentration aluminum sulfate using the formula below:
Concentration = number of moles ÷ volume. (1).
Recall; Number of moles = 3039.0664/ 342.15 = 8.8823 moles. The volume = 2050 gallons = 7759.25 Litres.
Therefore, slotting in the values of of number of moles and volume into the equation (1) above, we have;
Concentration= 8.8823 moles/ 7759.25 Litres. = 0.001145 M.
The next equation we are going to make use of today is that of the solubilty product that is;
Ksp = (Al^3+) (OH^-)^3.
So, we have the ksp = 1.3 × 10^-33 and the value of (Al^3+)= 0.001145 M.
Hence, making (OH^-) the subject of the formula;
(OH^-) = ( 1.3 × 10^-33 / 0.001145)^1/3.
(OH^-) = 8 × 10^-11 M.
Hence, pOH = - log (OH^-)
pOH = - log (8 × 10^-11).
pOH = 10.1
Therefore, the values of pH is; 1
pH + pOH = 14.
pH = 14 - 10.1 = 3.9.
Ответ:
Explanation:
Density is given by
The volume of acetic acid is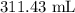 .
.