maguilarz2005
28.04.2021 •
Chemistry
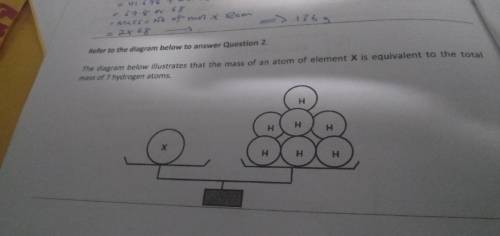
The mass of an atom of element x is equivalent to the total mass of 7 hydrogen atoms
Solved
Show answers
More tips
- O Other How to Accidentally Get a Rare Coin with Your Change and How to Know Its Value?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- D Dating, Love, Relationships Why do we feel shame?...
- F Family and Home Why Does God Punish Us Constantly and How Can We Fix It?...
- F Family and Home Do Lullabies Help Babies Sleep or Is it Just a Myth?...
- B Business and Finance How to Get Your Money Back When Lending? Top 7 Ways...
- S Society and Politics Выборы: Смысл, Значение и Отражение...
- D Dating, Love, Relationships Why Should the Man be Active and the Woman Passive during Foreplay?...
- P Philosophy How did the concept of module arise in computer science?...
- S Society and Politics Why are thugs called gopniks ? A fascinating journey through Russian subculture...
Answers on questions: Chemistry
- C Chemistry How many grams of oxygen are there in 15.5 grams of glucose, C6H12O6?...
- C Chemistry Unsaturated compounds . do not readily react have a high activation energy require UV light to react are very reactive...
- C Chemistry How many neutrons are in atom of an element that has 16 protons, 16 electrons, and a mass of 40?...
- C Chemistry The day table below gives the temperature and pressure of four different gas samples, each in a 2-liter container. Which two samples contain the same total number of particles?...
- C Chemistry I need help in chem please I have no idea what todo...
- C Chemistry Which of these ingredients is important in making plastics?...
- C Chemistry 11) Predict the products of this reaction. CuSO4(aq) + 2KOH(aq) → A) Cu(OH)2(aq) + K2SO4(s) B) Cu(OH)2(s) + K2SO4(aq) C) CuOH(s) + K2SO4(aq) D) CuOH2(s) + K2SO4(aq)...
- C Chemistry What is the full form of HPC...
- M Mathematics How do you divide 1.29 by 6...
- H History What is the “crime of the Ages ?...

Ответ:
[Ar] 4s2, 3d3
Explanation:
This is due to the loss of electeon, hence more proton.