violetmeadow4
26.11.2019 •
Chemistry
The maximum allowable concentration of pb2+ ions in drinking water is 0.05 ppm (i.e., 0.05 g of pb2+ in 1 million grams of water). (a) what is the pb2+ concentration of an underground water supply at equilibrium with the mineral cerussite (pbco3) (ksp = 3.3 × 10−14) ? × 10 g / lenter your answer in scientific notation. (b) does this concentration exceed the allowable concentration guideline? yes no
Solved
Show answers
More tips
- S Science and Technology The Metric System in Our Daily Life: Understanding Its Importance...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- C Computers and Internet How to Learn to Type Fast?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
- H Health and Medicine What is Autism? Understanding the Basics of This Neurodevelopmental Disorder...
Answers on questions: Chemistry
- C Chemistry How many liters are in one kiloliter?...
- C Chemistry What are two things that you observed when you heated the mixture of tin and nitric acid over the bunsen burner in the virtual lab, and why was it necessary to heat the mixture...
- C Chemistry A hydrocarbon with this formula C4H6: is chemically inert is somewhat reactive is unstable indeterminate...
- M Mathematics WILL GIVE BRAINLIEST...
- B Biology Which precautions are necessary for proper decomposition of domestic waste?...
- C Chemistry What is the strongest acid and strongest base found in water...
- M Mathematics 100 ÷ 100 + 12 + 1000is...
- C Computers and Technology WIRELESS DATA TRANSMISSION METHODS...
- M Mathematics Paul organised an event for a charity. Each ticket for the event cost £19.95 Paul sold 395 tickets. Paul paid costs of £6000 He gave all money left to the charity Work out the...
- E English Associated Riskin a crowded places...

Ответ:
a. 0,049g of Pb²⁺ in 1 million grams of water.
b. No
Explanation:
The equilibrium of PbCO₃ in water is:
PbCO₃ ⇄ Pb²⁺ + CO₃²⁻
Where ksp is defined as:
ksp = [Pb²⁺] [CO₃²⁻] (1)
The initial concentration of PbCO₃ in molarity is:
a. The concentrations in equilibrium are:
[PbCO₃} = 0,0374M-x
[Pb²⁺] = x
[CO₃²⁻] = x
Replacing in (1)
3,3x10⁻¹⁴ = x²
x = 1,82x10⁻⁷M
Thus, [Pb²⁺] = 1,82x10⁻⁷M, this value in g of Pb²⁺ per million grams of water is:
1,82x10⁻⁷M× ×
×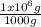 = 0,049 g of Pb²⁺ in 1 million grams of water
= 0,049 g of Pb²⁺ in 1 million grams of water
As this value is lower than the maximum allowable concentration of Pb²⁺, the concentration doesn't exceed this maximum value
I hope it helps!
Ответ:
Explanation:
The formula for a trapezoid is .
So once we put in the given data from the image, that gives us .
First, add the numerators.
16.5+6.5= 23
Now, divide the fraction.
23/2= 11.5
Finally, multiply.
11.5×7= 80.5