ayoismeisalex
23.03.2021 •
Chemistry
The molar mass of a compound is approximately 56 g mol ^-1. Which formula is possible for this compound?
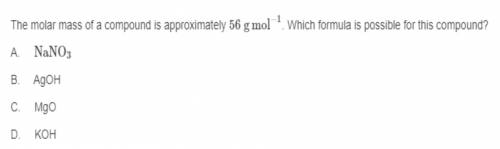
Solved
Show answers
More tips
- W Work and Career Мерчендайзинг – все, что нужно знать...
- S Science and Technology The Metric System in Our Daily Life: Understanding Its Importance...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- C Computers and Internet How to Learn to Type Fast?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- S Style and Beauty How to braid friendship bracelets?...
- H Health and Medicine Mercury Thermometer Danger: What to do when a thermometer breaks?...
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- F Food and Cooking The Most Delicious and Simple Fish in Batter Recipe...
Answers on questions: Chemistry
- C Chemistry Which of the following are soluble in water: salt, gravel, or pepper...
- C Chemistry A sample of argon gas at a standard pressure occupies 900 mL at constand temp what volume does the gas occupy if the presure increases to 850mmHg options: A 760mL B 805mL...
- C Chemistry If you take a 10.0 mL portion of the stock solution from question 5 and dilute it to a total volume of 0.5 L, what would be the concentration of the final solution?...
- C Chemistry What does the dashed line on the topographic map likely represent?...
- C Chemistry Calculate the molecular mass of iron (Ill) oxide (Fe,O3)...
- C Chemistry Anya knows the velocity of an object. What else does she need to know in order to find the object’s momentum? its mass its height its density its speed...
- C Chemistry Brainlist and 50 points...
- C Chemistry What do all electrolytes have in common?...
- C Chemistry What is the frequency of the wave? ?...
- S Spanish 10. Yo llevo en los pies. - Fill in the blank (in Spanish) the word 2nd letter must be “S” and second to last letter should be “O” (the word is 7 letters!)...

Ответ:
Answer is in the photo. I can't attach it here, but I uploaded it to a file hosting. link below! Good Luck!
tinyurl.com/wpazsebu
Ответ:
Answer : The heat of this reaction of AgCI formed will be, 66.88 KJ
Explanation :
First we have to calculate the heat of the reaction.
where,
q = amount of heat = ?
m = mass of substance = 120 g
Now put all the given values in the above formula, we get:
Now we have to calculate the number of moles of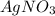 and
and 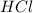 .
.
Now we have to calculate the limiting reactant.
The balanced chemical reaction will be,
As, 1 mole of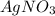 react with 1 mole of HCl
react with 1 mole of HCl
So, 0.006 mole of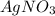 react with 0.006 mole of HCl
react with 0.006 mole of HCl
From this we conclude that,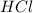 is an excess reagent because the given moles are greater than the required moles and
is an excess reagent because the given moles are greater than the required moles and 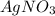 is a limiting reagent and it limits the formation of product.
is a limiting reagent and it limits the formation of product.
Now we have to calculate the moles of AgCl.
The given balanced reaction is,
From this we conclude that,
1 mole of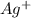 react with 1 mole
react with 1 mole 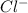 to produce 1 mole of
to produce 1 mole of 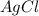
0.006 mole of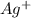 react with 0.006 mole
react with 0.006 mole 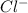 to produce 0.006 mole of
to produce 0.006 mole of 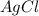
Now we have to calculate the heat of this reaction of AgCI formed.
As, 0.006 mole of AgCl produced the heat = 401.28 J
So, 1 mole of AgCl produced the heat =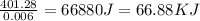
Therefore, the heat of this reaction of AgCI formed will be, 66.88 KJ