The multistep smelting of ferric oxide to form elemental iron occurs at high temperatures in a blast furnace. In the first step, ferric oxide reacts with carbon monoxide to form Fe₃O.₄ This substance reacts with more carbon monoxide to formiron(II) oxide, which reacts with still more carbon monoxide to form molten iron. Carbon dioxide is also produced in each step.
(a) Write an overall balanced equation for the iron-smelting process.
(b) How many grams of carbon monoxide are required to form 45.0 metric tons of iron from ferric oxide?
Solved
Show answers
More tips
- C Computers and Internet How to Choose an Uninterruptible Power Supply (UPS) for Your Computer: Expert Tips...
- F Food and Cooking The Health Benefits of Flaxseed oil...
- S Style and Beauty Why is Sugaring Better than Waxing for Hair Removal?...
- W Work and Career Where can you learn to be a flight attendant?...
- G Goods and services How to Properly Calculate the Power of Your Air Conditioner?...
- F Food and Cooking Effective Methods to Organize Videos in your iPad According to Content...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
Answers on questions: Chemistry
- C Chemistry 2 crassify the given quantities into solo quantity and vector quantity Area, velocity weight speed displacen dictance massa...
- C Chemistry Draw styrenedraw the structure of cyrene ...
- C Chemistry Can someone help me with Problem 21.100 ? Many thanks?...
- C Chemistry What is perodic table list all the elements up to 30 with their valencies...
- M Mathematics The most efficient first step in the process to factor the trinomial 4x^3-20x^2+24x a. factor out -1 b. factor out 4 c. factor out 4x d. factor out (x-3)...
- H History Match the inventor with the invention. 1. thomas savery invented the spinning jenny 2. james watt built a steamship, the clermont 3. james hargreaves built steam...
- S Spanish 5(-2n+4)+2(n+3) quiero saber la respuesta...
- B Biology Can you answer all of these questions...
- M Mathematics Mandy is setting up for a reception. for the number of seats needed, she knows she can use either 8 seats at each table with none leftover or 10 seats at each table...
- M Mathematics Abag contains 10 blue, 6 green, and 4 red marbles. you choose a marble with one hand and then a second marble with the other hand. what is the probability that...

Ответ:
a)
b) 33749996 grams
Explanation:
a) The balanced chemical reactions will be :
Overall equation :
Overall balanced equation :
b) Amount of iron = 45 metric ton = 45000 kg = 45000000g
To calculate the moles :
According to stoichiometry :
2 moles of are produced from= 3 moles of
are produced from= 3 moles of 
Thus 803571 moles of are produced from=
are produced from= of
of 
Mass of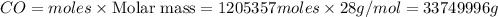
Thus 33749996 g of carbon monoxide are required to form 45.0 metric tons of iron from ferric oxide
Ответ: