The net ionic equation for the reaction that occurs during the titration of nitrous acid with sodium hydroxide is (a) hno2 na 1 oh-1 nano2 h2o (b) hno2 naoh na 1 no2-1 h2o (c) h 1 oh-1 h2o (d) hno2 h2o no2-1 h3o 1 (e) hno2 oh-1 no2-1 h2o
Solved
Show answers
More tips
- S Style and Beauty How to Get Rid of a Double Chin?...
- F Food and Cooking How to Cook Julienne? Recipes and Tips...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- F Food and Cooking Apple Cider Vinegar: The Ultimate Health and Beauty Solution...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
Answers on questions: Chemistry
- C Chemistry How does a multicellular organism develop? the cells shrink. one cell grows bigger. the cells replicate to form exact copies. the cells differentiate....
- C Chemistry Which of these statements is correct? eukaryotic cells do not have ribosomes. eukaryotic cells do not have chloroplasts. prokaryotic cells do not have a cell wall. prokaryotic cells...
- C Chemistry The oxidation number of hydrogen when it is in a compound other than a hydride is (1 point...
- C Chemistry Calculate the concentration in grams per mL of a 450.0 mL solution containing 0.0762 moles of iodine. Work please...
- C Chemistry What volume of solution would be needed to deliver 3.0 mg of a drug if the concentration of the drug in the solution was 3.5% (m/v)? Show work please...
- C Chemistry Calculate ΔS for the isothermal compression of 3.05 mol of Cu(s) from 1.00 bar to 1370. bar at 298 K. α=0.492×10−4K−1,κT=0.78×10−6 bar−1, and the density is 8.92 g⋅cm−3....
- C Chemistry Spongebob was trying to make Krabby Patties that weighed 12oz. He made 4 Krabby Patties. They all had the following weights: 12.12oz; 11.9oz; 12.2oz; 12.1oz. Is this an example of...
- C Chemistry What is the independent variable? Picture...
- C Chemistry Which of the following choices is NOT a consequence of incomplete combustion? A. Carbon monoxide blocking oxygen in the blood. OB. Carbon particles making buildings dirty. C. Carbon...
- C Chemistry How could you in vestigate the pollution of Groundwater...

Ответ:
Explanation:
Complete ionic equation : In complete ionic equation, all the substance that are strong electrolyte and present in an aqueous are represented in the form of ions.
Net ionic equation : In the net ionic equations, we are not include the spectator ions in the equations.
Only the species which are present in aqueous state dissociate.
Spectator ions : The ions present on reactant and product side which do not participate in a reactions. The same ions present on both the sides.
(a)
The balanced molecular equation will be,
The complete ionic equation in separated aqueous solution will be,
In this equation the species present are, are the spectator ions.
are the spectator ions.
Hence, the net ionic equation contains specie is
Ответ:
The net ionic equation for the reaction has been,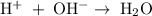 .
.
Thus, option C is correct.
The net ionic equation has been given as formation of ions involved in the reaction and the formed products.
Net Ionic EquationThe titration of nitrous acid and sodium hydroxide has been given in chemical equation as:
The ionic equation has been:
In the net ionic equation, the ions same on the product and reactant side are eliminated.
Thus, the net ionic equation for the reaction has been:
Thus, option C is correct.
For more information about net ionic equation, refer to the link:
link
Ответ:
dad is that u
Dude c.u.m now
Explanation: