snowprincess99447
06.09.2019 •
Chemistry
The percent chloride in an unknown sample may be determined by gravimetric methods. suppose a 0.6789g sample of an unknown chloride sample was dissolved and agcl is precipitated by adding agno3 solution. the precipitate was filtered, ignited, and found to weigh 0.g. what was the percent chloride in the sample?
Solved
Show answers
More tips
- F Food and Cooking How to Choose the Right Olive Oil: A Comprehensive Guide...
- P Philosophy Is Everything We Strive for Eventually Achieved and Destroyed?...
- S Society and Politics Understanding Politics and Its Role in the Development of Civilization...
- P Philosophy Why Did God Create Man and Place Him in Obscurity?...
- S Society and Politics Skoptsy: Who They Are and How They Perform Castration?...
- O Other Childhood Fears: What Many of Us Experienced...
- P Philosophy What is Something for you?...
- H Health and Medicine Why Do Humans Have One Heart?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- O Other How to Accidentally Get a Rare Coin with Your Change and How to Know Its Value?...
Answers on questions: Chemistry
- C Chemistry How much work is accomplished when a force of 250 n pushes a box across the floor for a distance of 50 meters? a. 5 n b. 5 j c. 12,500 j d. 12,500 n...
- C Chemistry Which of the following is the largest disadvantage of hydropower?...
- C Chemistry What is the correct order to form a glucose molecule...
- C Chemistry Vinegar, hcl, and h2so4 are examples of (4 points) salts acids bases ionic compounds...
- C Chemistry Calculate the mass of 3.05 buckets of golf balls....
- C Chemistry When the pH of an aqueous solution is changed from 1 to 2, the concentration of hydronium ions in the solution...
- C Chemistry The pressure of a sample of gas of constant volume is 2.0 atm at 30.ºC. What is the pressure of this sample at 20.ºC?...
- C Chemistry A magnet is surrounded by , which applies a push or a pull to an object without actually touching it....
- C Chemistry There are 3.0 g of NO and 3.0 g of CO reacting. what is the limiting reactant ?...
- C Chemistry What colour would stomach acid to turn universal indicator?...

Ответ:
The percent chlorine in the sample is 12.08 %.
Explanation:
We are given:
Mass of AgCl precipitated = 0.3333 g
Molar mass of AgCl = 143.32 g/mol
Molar mass of Chlorine atom = 35.45 g
In 1 mole of AgCl, 1 mole of chlorine atom is present.
In 143.32 grams of AgCl, 35.45 g of chlorine atom is present.
So, in 0.3333 grams of AgCl, the mass of chlorine present will be =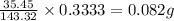
To calculate the percentage composition of chlorine in sample, we use the equation:
Mass of sample = 0.6789 g
Mass of chlorine = 0.082 g
Putting values in above equation, we get:
Hence, the percent chlorine in the sample is 12.08 %.
Ответ:
B
Step-by-step explanation: