brainlord4209
11.02.2021 •
Chemistry
The reaction heated up 100 g of water in the beaker from 25 °C to 50.0 °C. The specific heat capacity of water is 4.184 J/g°C. Calculate the amount of heat energy that was released by the reaction.
Solved
Show answers
More tips
- L Leisure and Entertainment How to Learn to Draw Graffiti: Tips for Beginners...
- P Photography and Videography How to Choose a Digital Camera?...
- C Computers and Internet How to Properly Order Clothing from International Online Stores...
- F Food and Cooking How to Calculate the Gender of Your Child with Blood?...
- S Society and Politics 10 Tips for Boosting Your Self-Esteem...
- C Computers and Internet How to Create a Folder on Your iPhone?...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- E English Juliet threatens to kill herself if Friar Lawrence doesn t -Help her out -Help her escape to Mantua -Somehow get her out of this marriage with Paris -Help her abandon her family...
- E English Subordinating conjuctuon jigsaws. The arrogant MAN sneered...
- P Physics Adeer with a mass of 146kg is running head on toward you with a speed of 17 m/s. you are going north. find the momentum of the deer ....

Ответ:
The amount of heat energy that was released by the reaction is 10,460 J.
Explanation:
Sensible heat is that which a body or an object receives or releases and causes its temperature to increase or decrease without affecting its molecular structure and therefore its state.
In other words, sensible heat is the amount of heat that a body absorbs or releases without any changes in its physical state (phase change). When a body is supplied or absorbed sensible heat, the temperature varies.
The equation that allows calculating heat exchanges is:
Q = c * m * ΔT
where Q is the heat exchanged by a body of mass m, made up of a specific heat substance c and where ΔT is the temperature variation.
In this case:
c= 4.184Replacing:
Q= 4.184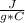 *100 g* 25 C
*100 g* 25 C
Solving:
Q=10,460 J
The amount of heat energy that was released by the reaction is 10,460 J.
Ответ:
Explanation:
your good bro