The reaction of methyl iodide with sodium azide, NaN3, proceeds by an SN2 mechanism. What is the effect of doubling the concentration of NaN3 on the rate of the reaction?
Solved
Show answers
More tips
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
- F Food and Cooking Delight for Gourmets: How to Prepare Liver Pate...
- C Computers and Internet How to Learn to Type Fast?...
- H Health and Medicine Angina: Causes, Symptoms, and Treatment...
- D Dating, Love, Relationships How to Overcome Jealousy: Tips and Tricks...
- H Health and Medicine 10 Ways to Cleanse Your Colon and Improve Your Health...
- W Work and Career How to Start Your Own Business: Tips and Recommendations...
- F Food and Cooking How to Make Delicious Cabbage Pies: The Best Recipes!...

Ответ:
Rate will increase by a factor of 2.
Explanation:
As the reaction proceeds by an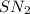 mechanism, both the reactants take part in the reaction and the rate law for the reaction will be:
mechanism, both the reactants take part in the reaction and the rate law for the reaction will be:
Thus when concentration of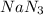 is doubled ,
is doubled ,
Thus the rate of the reaction would increase by a factor of 2.
Ответ:
It’s a and e
Explanation: