The Solubility Product Constant for calcium bromide is > 1. If calcium bromide is dissolved in water you can say that the equilibrium concentrations of calcium and bromide ions are: ... ...A. High ...B. Moderate ...C. Low The solubility of calcium bromide in water is: ... ...A. High ...B. Moderate ...C. Low
Solved
Show answers
More tips
- C Computers and Internet Отправляем смс через интернет: легко и просто...
- H Health and Medicine Want to Lose Weight? Here s How Many Calories You Need Per Day...
- H Health and Medicine How Many Ribs Do Humans Have?...
- C Computers and Internet Dropbox: What is it and How to Use it...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- A Auto and Moto How many blood alcohol level units are allowed in Russian traffic laws?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- H Health and Medicine How to Deal with Heat Stroke?...
- H Health and Medicine Sunstroke: Causes, Symptoms, and Precautions...
Answers on questions: Chemistry
- C Chemistry What would happen if you mix an acid and a base?...
- C Chemistry In which of the following orbitals is the fifth valance electron in a fluorine molecule situated?...
- C Chemistry ITEM BANK: Move to Bottom 1 2 3 5 6 7 12 drag and drop answer here C2H6+ 702 6H2O + 4CO2 drag and drop answer here HCIO4 + P4010 4H3PO4 + drag and drop answer here...
- C Chemistry Given its formula and Avogadro s Number (6.02 x 10^23 molecules/mol), deduce how many molecules are present in 3 x 10^-16 grams of TCDD. Type in only a number without...
- C Chemistry Children s Tylenol contains 32 mg of acetaminophen per milliliter of a liquid. The recommended dose of this medicine for a 50-pound child is 2.0 teaspoons. How many...
- C Chemistry Aniline involves an amine, which is a Choose... functional group. When an aqueous acid solution is added to an organic solution including aniline, the aniline appears...
- C Chemistry Help! Can somebody explain the answer and how they got it? Write the equation for the first ionization of potassium....
- M Mathematics Someone help! I’m not good at these problems!...
- A Arts My discord is valentina05#8885 add me...
- C Chemistry Can someone please help me please?...

Ответ:
(1). If calcium bromide is dissolved in water you can say that the equilibrium concentrations of calcium and bromide ions are high.
(2). The solubility of calcium bromide in water is high.
Explanation:
We know that for a reaction its solubility product is equal to the product of concentration of ions present.
For example,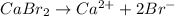
As it is given that for this reaction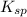 > 1. Hence, the product of concentration of both these ions will be greater than 1 which means that products are favored.
> 1. Hence, the product of concentration of both these ions will be greater than 1 which means that products are favored.
Therefore, equilibrium concentrations of calcium and bromide ions are high.
Also, more is the number of these ions formed more will be the solubility of the compound (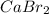 ).
).
As a result, the solubility of calcium bromide in water is high.
Ответ:
The work done on neon = -323 J
The internal energy change= -392.84 J
The heat absorbed by neon = -69.84 J
Explanation:
Step 1: Data given
Number of moles = 0.500 moles
Pressure = 1 atm
Temperature = 273 Kelvin
The pressure will change from 1.00 atm to 0.200 atm. The temperature changes from 273 to 210 Kelvin.
a) calculate the work done on neon
W = -P(V2-V1)
⇒ with P = the pressure = 0.1 atm
⇒ with V1 = the initial volume = nRTi /Pi
⇒ with V2 = the final volume = nRTf /Pf
W = -PnR((T2/P2) -(T1/P1))
⇒ with T2 = the final temperature = 210 K
⇒ with T1 = the initial temperature = 273 K
⇒ with P2 = the final pressure = 0.200 atm
⇒ with P1 = the initial pressure = 1.00 atm
W = -nR (210*(0.1/0.2) - 273*(0.1/1.00))
W = -nR*(105 - 27.3)
W= -(0.500)*(8.314)*(77.7)
W = -323 J
b) calculate the internal energy change
E = (3/2)*nRT
ΔE = Ef - Ei
ΔE =(3/2)*nR(T2-T1)
⇒ with n= number of moles = 0.500 moles
⇒ with T2 =the final temperature = 210 K
⇒ with T1 = the initial temperature = 273 K
ΔE = (3/2)*(0.5)*(8.314)(210-273)
ΔE = -392.84 J
c) Calculate the heat absorbed by neon
ΔE = q + W
q = ΔE -W
⇒ with ΔE = -392.84 J
⇒ with W = -323 J
q = -392.84 J -( -323 J)
q =-392.84 J + 323 J
q = -69.84 J