The standard molar heat of fusion of ice is 6020 j/mol. calculate q, w, and ∆e for melting 1.00 mol of ice at 0◦c and 1.00 atm pressure. 1. q = 6020 j/mol
Solved
Show answers
More tips
- F Food and Cooking What can and cannot be eaten during Lent?...
- F Food and Cooking How to Find Your Zip Code?...
- W Work and Career Can Skill Alone Make You a Professional?...
- C Computers and Internet How to Top Up Your Skype Account Without Losing Money?...
- P Philosophy Unidentified Flying Object - What is the Nature of this Phenomenon?...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
Answers on questions: Chemistry
- C Chemistry Describe the movement of air over two nearby land areas, one of which is heated more than the other...
- C Chemistry Where would a boat produce the highest concentration of carbon monoxide? on the starboard?...
- C Chemistry Which if the following is the term for the phase change from gas to water...
- C Chemistry How did the concentration of iodide ions change in these two trials, how did the rate change accordingly?...
- C Chemistry Four type of molecular models...
- C Chemistry The range in size of most atomic radii is approximately is 5 × 10 −11 m to 2 × 10 −10 m. but why is it that and how?...
- C Chemistry Which type of mutation can contribute to the development of many types of cancers...
- C Chemistry What type of bond will form if an element with a electro negativity of 3.1 bonds to an element of electro negativity of 0.9...
- C Chemistry What is the answer i need help...
- C Chemistry What are the uses of hydrogen? ...

Ответ:
Answer : q = 6020 J, w = -6020 J, Δe = 0
Solution : Given,
Molar heat of fusion of ice = 6020 J/mole
Number of moles = 1 mole
Pressure = 1 atm
Molar heat of fusion : It is defined as the amount of energy required to melt 1 mole of a substance at its melting point. There is no temperature change.
The relation between heat and molar heat of fusion is,
or,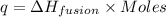 (in terms of moles)
(in terms of moles)
Now we have to calculate the value of q.
When temperature is constant then the system behaves isothermally and Δe is a temperature dependent variable.
So, the value of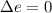
Now we have to calculate the value of w.
Formula used :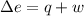
where, q is heat required, w is work done and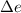 is internal energy.
is internal energy.
Now put all the given values in above formula, we get
w = -6020 J
Therefore, q = 6020 J, w = -6020 J, Δe = 0
Ответ:
Trial 1 (20.61 mL HCl):
⇒ 0.121 M
Trial 2 (20.06 mL HCl):
⇒ 0.125 M
Trial 3 (19.67 mL HCl):
⇒ 0.122 M
the answer for the next question, Compute the average concentration (molarity) of HCl for all three trials, is 0.123 M.
Explanation: