susanspagoot6385
12.07.2019 •
Chemistry
The sulfur dioxide (so2) stack-gas concentration from fossil-fuel combustion is 12 ppmv. determine the stack-gas so2 concentration in units of µg/m3. assume 0 °c and 1 atm pressure. express answer to nearest unit.
Solved
Show answers
More tips
- G Goods and services How to Choose a Coffee Maker? The Ultimate Guide for Coffee Lovers...
- O Other How to Accidentally Get a Rare Coin with Your Change and How to Know Its Value?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- D Dating, Love, Relationships Why do we feel shame?...
- F Family and Home Why Does God Punish Us Constantly and How Can We Fix It?...
- F Family and Home Do Lullabies Help Babies Sleep or Is it Just a Myth?...
- B Business and Finance How to Get Your Money Back When Lending? Top 7 Ways...
- S Society and Politics Выборы: Смысл, Значение и Отражение...
- D Dating, Love, Relationships Why Should the Man be Active and the Woman Passive during Foreplay?...
- P Philosophy How did the concept of module arise in computer science?...

Ответ:
The concentration of in the stack gas = 12 ppmv
in the stack gas = 12 ppmv
That means 12 L of is present per
is present per 
The given temperature is 273 K (0 C) and pressure is 1 atm. At these conditions, 1 mol of gas would occupy,
V = 22.4 L
1 mol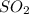 occupies 22.4 L
occupies 22.4 L
Moles of =
= 
Mass of =
= =
= μg
μg
Converting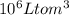 :
:
Calculating the concentration in μg/ :
:
Ответ: